| Posted: Nov 13, 2012 | |
Tracking nanomedicines inside the body |
|
| (Nanowerk Spotlight) One of the key issues in the development of novel nanomedicines is the ability to track nanoscale drug carriers inside the body to evaluate where they go and how they get there. In order to get to products that get regulatory approval and can be administered to patients, scientists need to come up with noninvasive test for the biodistribution of nanomedicines. | |
| "Virtually all previous preclinical studies in this area of research rely on 2D Fluorescence Reflectance Imaging (FRI)," Dr. Twan Lammers, a researcher at the Department of Experimental Molecular Imaging, Helmholtz Institute for Biomedical Engineering, RWTH-Aachen University in Germany, tells Nanowerk. "Given the limitation of 2D FRI in not being able to detect the fluorescence in deep-seated organs and tissues, 3D Fluorescence Molecular Tomography (3D FMT) emerged as an alternative. However, the lack of anatomical information – to assign the fluorescence signal to specific organs – was an important barrier hindering the routine use of standard 3D FMT for in vivo imaging of nanomedicines." | |
| Lammers adds that it should be noted that this situation is very different for activatable probes used as molecular diagnostics, which were already used relatively extensively in conjunction with 3D FMT, and for which Ale, Ntziachristos and colleagues also recently reported a hybrid CT-FMT setup ("FMT-XCT: in vivo animal studies with hybrid fluorescence molecular tomography–X-ray computed tomography"). | |
| Such a hybrid CT-FMT-based imaging protocol to enable more meaningful and more quantitative in vivo analyses has now been developed by a team, led by Lammers, that involved scientists from Aachen University, University of Twente, Utrecht University, and the Academy of Sciences of the Czech Republic. | |
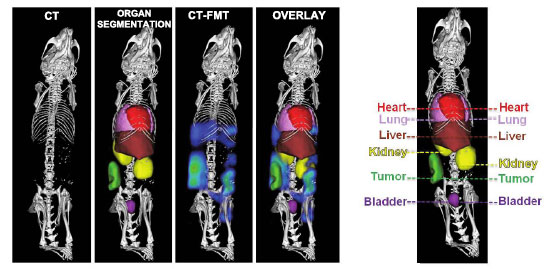 |
|
| Fig. 1: CT-based organ segmentation and hybrid CT-FMT imaging. High-resolution µCT scans of CT26 colon carcinoma-bearing nude mice, depicting highly electron-dense anatomical structures (i.e., bones), presegmented organs, FMT-based biodistribution data overlaid on highly electron-dense anatomical structures, and FMT-based biodistribution data overlaid on presegmented organs. Right panel: Two-dimensional planes representing individual organs. (Reprinted with permission from American Chemical Society) | |
| Reporting their findings in the October 15, 2012 online edition of ACS Nano ("Noninvasive Optical Imaging of Nanomedicine Biodistribution"), the team was able to visualize and quantify the biodistribution and target site accumulation of nanomedicines in mice using hybrid 3D CT-FMT imaging. | |
| Specifically, the team used computer tomography to enable a more accurate analysis of probe localization in non-superficial organs – something that is impossible using 2D FRI and standard 3D FMT. | |
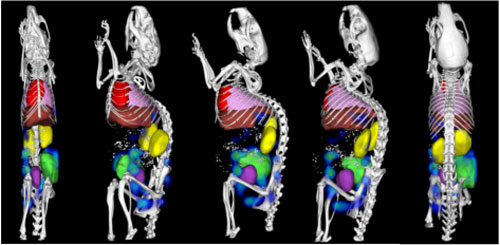 |
|
| Fig. 2: The overlay image from Fig. 1 in various orientations. (Image: Dr. Lammers, Helmholtz Institute for Biomedical Engineering) | |
| "Given the fact that optical imaging is ever more extensively used to monitor the in vivo behavior of nanomedicines, our findings therefore advance the field by enabling the visualization and quantification of probe accumulation of besides subcutaneous tumors also in a number of deeper-seated healthy tissues," says Lammers. | |
| Fluorescence molecular tomography is a technique which enables quantitative imaging of the fluorescent probe, or marker concentration to the picomolar level with no tissue depth limitation. Lasers are used to excite near-infrared probes for which planar detectors such as CCD cameras record excitation and emission images of the diffuse light propagation. Computer software then uses advanced algorithms to volumetrically reconstruct the accumulation in and the concentration of the optical imaging agents. | |
| So far, though, the major shortcoming of FMT has been its inability to accurately assign the reconstructed probe accumulation signal to a given organ of interest. | |
| The team's hybrid imaging protocol, a combination of high-resolution microcomputed tomography and FMT, enables more meaningful and more quantitative in vivo analyses of the biodistribution of nanomedicines. | |
| As Lammers points out, this initial proof-of-principle study solves one of the most important limitations which researchers working on the optical imaging of nanomedicine biodistribution have been confronted with, i.e. the inability to non-invasively and accurately assess probe accumulation in non-superficial organs. | |
| Hybrid 3D CT-FMT imaging can be used at the preclinical levels for a large number of in vivo applications, ranging from the molecular diagnosis of receptor expression or enzyme activity to the longitudinal visualization and quantification of drug delivery and drug release. | |
| The researchers caution that there is still quite some room for improvement and optimization of this technique. | |
| "Particularly" says Lammers, "the volumetric fluorescence reconstruction can be improved by including tissue-specific optical properties such as absorption and scattering in the reconstruction method. Furthermore, we are working on methods to perform automated organ segmentation to reduce the required effort per scan and achieve higher user independence." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
