| Posted: Jan 08, 2013 | |
Nanomaterial platform captures and releases circulating tumor cells |
|
| (Nanowerk Spotlight) Metastasis – the process of cancer spreading from its original site to distant tissues – is caused by marauding tumor cells that break off from the primary tumor site and ride in the bloodstream to set up colonies in other parts of the body. These breakaway cancer cells in the peripheral blood are known as circulating tumor cells (CTCs). Detecting and analyzing these cells can provide critical information for managing the spread of cancer and monitoring the effectiveness of therapies (see our previous Nanowerk Spotlight: "Capturing circulating cancer cells could provide insights into how disease spreads"). | |
| As important as the castaway cells are, scientists don't know a lot about them. They are rare, at about one CTC per billion blood cells. And they are not all identical, even if they come from the same tumor. | |
| In new work, researchers have created a Velcro-like platform which can not only capture CTCs with very high efficiency, but also release them in order to biochemically analyze them further. The team, led by Hsiao-hua Yu from the RIKEN Advanced Science Institute in Japan and Hsian-Rong Tseng from the Department of Molecular and Medical Pharmacology at the University of California Los Angeles, reports their new platform in the December 17, 2012 online edition of Advanced Materials ("Capture and Stimulated Release of Circulating Tumor Cells on Polymer-Grafted Silicon Nanostructures"). | |
| "Similar cell-capture devices have been reported before but our technology is unique in that it is capable of catching the tumor cells with great efficiency and releasing them with great cell viability," Yu tells Nanowerk. "This will make it possible to use CTCs for 'liquid biopsies' in the areas of cancer diagnostics and cancer management." | |
| Blood is passed through the device like a filter that contains a molecule capable of adhering to tumor cells like Velcro and separating them with efficiency ranging from 40% to 70%. The CTCs are retained by tiny temperature-responsive polymer brushes inside the device. | |
 |
|
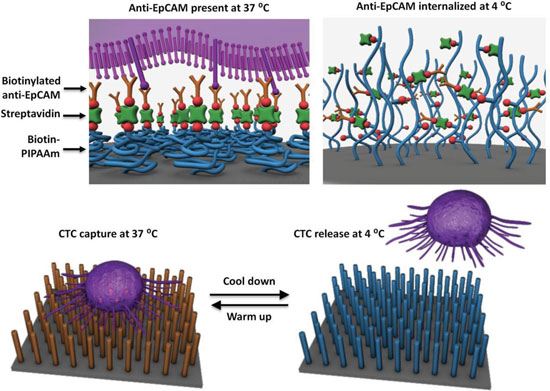
| Conceptual illustration of a new-generation nanomaterial-based platform for cell-affinity assay capable of not only capturing CTCs with superb efficiency, but also releasing upon stimulation these captured CTCs at a reduced temperature. (Reprinted with permission from Wiley-VCH Verlag) | |
| The novelty of this device is its release mechanism. At body temperature – 37 degrees Celsius – these polymer brushes stick to the tumor cells, but when cooled to 4 degrees Celsius, they release them, allowing scientists to examine the cells with biochemical methods. | |
| In order to confer thermal responsiveness onto their 'nano Velcro', the team covalently grafted biotin-functionalized polymer brushes (i.e., PIPAAm) onto a silicon nanowire substrate. At 37°C, the biotin groups and hydrophobic domains of biotinylated PIPAAm are present on the surfaces. | |
| "Via biotin-streptavidin interaction, biotinylated anti-EpCAM can be introduced, enabling highly efficient CTC capture," explains Yu. "When the temperature is lowered to 4°C, the backbones of surface-grafted biotinylated PIPAAm expand, resulting the internalization of anti-EpCAM and releasing CTCs from the substrates." | |
| He points out that approximately 90% of the released cells remained viable and that they can be further expanded in culture. | |
| "It is conceivable that the CTC-derived molecular signatures and functional readouts obtained from such platform will provide valuable insight into tumor biology," concludes Yu. "Eventually, in therapeutic intervention used in nanotechnology in healthcare can be properly guided to make a significant difference during the critical window of tumor progression and metathesis." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
