| Posted: Mar 15, 2013 | |
D-carbon - a new carbon allotrope with extremely high gas adsorption and mechanical properties |
|
| (Nanowerk Spotlight) Carbon is the fourth-most-abundant element in the universe and, depending on the arrangements of carbon atoms, takes on a wide variety of forms, called allotropes. Carbon allotropes exhibit unique properties of strength and electrical conductivity. Solid carbon at room temperature has two classical structures: diamond and graphite. In 1985 the discovery of the existence of a third and new carbon allotrope containing sixty perfectly symmetrically arranged carbon atoms (C60 or fullerene) meant a major breakthrough and opened a novel field of carbon nanochemistry. Then, in 1991, carbon nanotubes were discovered and graphene in 2004. Now, a research group in China has designed a novel carbon allotrope they've named D-carbon. | |
| "The D-carbon framework holds a diamond-like geometrical structure, and it is formed by sp3-sp hybridized carbon atoms, which is different from the sp3-sp3 hybridization diamond, also different from the sp2-sp2 hybridization fullerene, carbon nanotube and graphene," Dapeng Cao, a professor at the State Key Laboratory of Organic-Inorganic Composites at Beijing University of Chemical Technology, tells Nanowerk. "Moreover, the brand-new carbon framework exhibits not only extremely high excess methane uptake (255 V(STP)/V at 298 K and 35 bar), but also high bulk modulus of 91.7 GPa, which is one order of magnitude larger than other porous materials like metal-organic frameworks (MOFs), covalent-organic frameworks (COFs) and porous aromatic frameworks (PAFs)." The researchers report their findings in a recent issue of Journal of Materials Chemistry A ("A porous diamond carbon framework: a new carbon allotrope with extremely high gas adsorption and mechanical properties "). | |
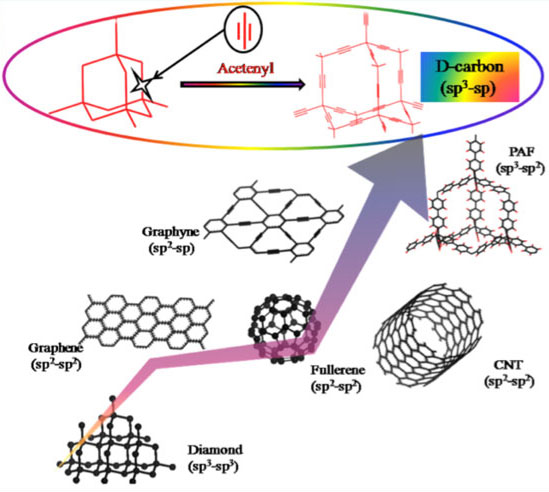 |
|
| Figure 1: The geometrical structures and carbon hybridization styles of D-carbon framework and other carbon allotropes. (Image: Prof. Cao, Beijing University of Chemical Technology) | |
| In previous publications, Cao and his collaborators have designed a porous aromatic framework with diamond-like structure (see "Targeted Synthesis of a Porous Aromatic Framework with High Stability and Exceptionally High Surface Area" and "High-Capacity Hydrogen Storage in Porous Aromatic Frameworks with Diamond-like Structure"). "Recently, by carbonizing the PAF materials, my collaborators and I found that the carbonized PAFs exhibit obvious improvement for gas adsorption (see "Selective adsorption of carbon dioxide by carbonized porous aromatic framework"), because the carbonization eliminates the excluded effect of hydrogen atom in phenyl rings on the guest, and enhances the affinity of the framework toward gas molecules," says Cao. "Therefore, we believed that an all-carbon framework would be advantageous for gas storage. Motivated by the points above, we designed the brand-new D-carbon framework and further predicted their properties." | |
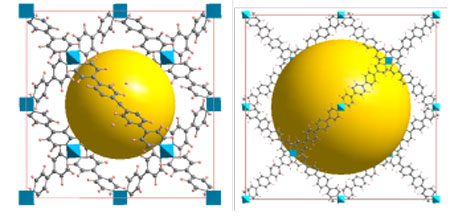 |
|
| Figure 2: Geometrical Structures of PAF-302 and PAF-304. (Image: Prof. Cao, Beijing University of Chemical Technology) | |
| The researchers designed D-carbon by inserting -CΞC- linkers into all the C-C bonds in a diamond. The C?C (sp3?sp) and CΞC (sp?sp) bonds in the D-carbon framework are of 1.53 Â and 1.22 Â, respectively. | |
| "We also calculated the cohesive and formation energies of D-carbon," says Cao. "Results indicate that D-carbon is energetically favored and thermodynamically stable for experimental synthesis." | |
| He notes that the D-carbon framework exhibits not only extremely high excess methane uptake, but also high bulk modulus. Therefore, it would be an excellent adsorption and high modulus material. Of course, once it can be reasonably functionalized, it would be a very versatile material that can be used as catalyst, drug delivery system and fuel cells etc. | |
| Of course, how to synthesize the D-carbon and how to obtain a perfect crystal are still a great challenge for future research in this area. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
