| Posted: Feb 23, 2006 | |
Pinpoint, on-demand drug delivery with nanotubes is now possible |
|
| (Nanowerk News) Researchers from the Martin Research Group at the University of Michigan have demonstrated they can precisely release individual drugs and bioactive molecules at desired points in time by using electrical stimulation of nanotubes. | |
| "There are many potential applications of our work" Professor Martin told Nanowerk "since we have demonstrated the ability to precisely control both the spatial and temporal delivery of drugs using the polymer nanotubes. This means the drugs now come out when we want them and where we want them." | |
| Martin's research provides a means for creating low impedance, biologically active polymer coatings, allowing the development of highly biocompatible electrodes that facilitate integration at the electrode -living tissue interface. Apart from precise drug-delivery, the potential biomedical applications of these molecule- eluting, electrically active polymer nanotubes could include highly localized stimulation of neurite outgrowth and guidance for neural tissue regeneration through functionalizing microelectrodes on neural prostheses. | |
| Conducting polymers are of considerable interest for researchers working on biomedical applications. Their response to electrochemical oxidation or reduction can produce a change in conductivity, color, and volume. Martin's group developed a method to prepare conducting-polymer nanotubes that can be used for precisely controlled drug release. | |
| The fabrication process involves electrospinning of a biodegradable polymer, into which a drug has been incorporated, followed by electrochemical deposition of a conducting-polymer around the drug-loaded, electrospun biodegradable polymers. The conducting-polymer nanotubes significantly decrease the impedance and increase the charge capacity of the recording electrode sites on microfabricated neural prosthetic devices. The drugs can be released from the nanotubes in a desired fashion by electrical stimulation of the nanotubes. | |
| A big problem with the implantation of microelectrodes and neural prosthetic devices in the nervous system is associated with tissue injury and inflammation. This results in neuronal loss near electrodes and encapsulation of the device in a high impedance barrier of glia and immune cells, undermining the goal of maintaining long-term communication with neurons. Minimizing the electrode impedance therefore is an important requirement for obtaining highquality signals (high signal-to-noise ratio). | |
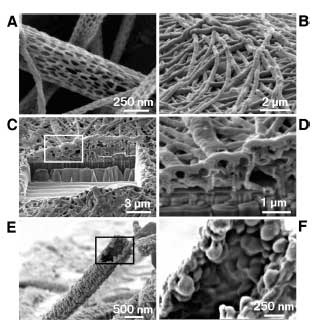 | |
| Scanning electron micrographs of PLGA nanoscale fibers and PEDOT nanotubes. A) diameters of the PLGA fibers were distributed over the range 40–500 nm with the majority being between 100–200 nm. B) Electropolymerized PEDOT nanotubes on the electrode site of an acute neural probe tip after removing the PLGA core fibers. C) A section of (B) cut with a FIB showing the silicon substrate layer and PEDOT nanoscale fiber coating. D) Higher-magnification image of (C) showing the PEDOT nanotubes crossing each other. E) A single PEDOT nanotube which was polymerized around a PLGA nanoscale fiber, followed by dissolution of the PLGA core fiber. This image shows the external texture at the surface of the nanotube. F) Higher-magnification image of a single PEDOT nanotube demonstrating the textured morphology that has been directly replicated from the external surface of the electrospun PLGA fiber templates. The average wall thickness of PEDOT nanotubes varied from 50–100 nm, with the nanotube diameters ranging from 100 to 600 nm. (Source: University of Michigan; Re-printed with permission of WILEY-VCH Verlag GmbH & Co. KGaA) | |
| Martin believes that both the biocompatibility and electrical signal transduction capacity of neural prosthetic devices and deep brain stimulators can be greatly enhanced by integration with the technology that they are developing. His studies have shown that PEDOT (the conducting polymer poly(3,4-ethylene dioxythiophene)) can be polymerized around living cells and tissues with little toxicity. This suggests that PEDOT networks can be grown within living tissue, for the purpose of extending electrodes to contact healthy neurons beyond the halo of dead cells and inflammatory tissue that encapsulates chronically implanted neural prosthetics. | |
| A variety of proteins, including Nerve Growth Factor, collagen, laminin, and poly(lysine) can be incorporated into the PEDOT matrix. Protein within the PEDOT is bioactive. It can be recognized by cells in contact with the PEDOT and can be passively or actively released from the polymer matrix. | |
| In their recent work, Martin describes how they prepared drug-loaded PEDOT nanotubes on the surface of a microelectrode. The process starts by electrospinning nanofibers of the biodegradable polymer PLGA (poly(lactide-co-glycolide)) onto the surface of a neural probe followed by electrochemical deposition of conducting polymers around the electrospun nanofibers. In a final step, the fiber templates can be removed or allowed to slowly degrade, providing additional means of controlled delivery of biologically active agents incorporated into the fibers themselves. | |
| The nanotubes deliver their bioactive contents either by passive delivery resulting from controlled degradation of the PLLA/PLGA or other matrix polymer used in the core or by actively actuating the drug-loaded nanotubes with an applied electrical field. | |
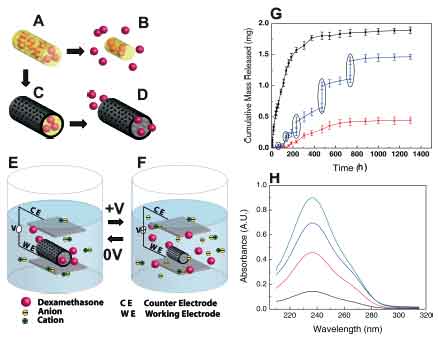 | |
| Schematic illustration of the controlled release of dexamethasone: A) dexamethasone-loaded electrospun PLGA, B) hydrolytic degradation of PLGA fibers leading to release of the drug, and C) electrochemical deposition of PEDOT around the dexamethasone-loaded electrospun PLGA fiber slows down the release of dexamethasone (D). E) PEDOT nanotubes in a neutral electrical condition. F) External electrical stimulation controls the release of dexamethasone from the PEDOT nanotubes due to contraction or expansion of the PEDOT. By applying a positive voltage, electrons are injected into the chains and positive charges in the polymer chains are compensated. To maintain overall charge neutrality, counterions are expelled towards the solution and the nanotubes contract. This shrinkage causes the drugs to come out of the ends of tubes. G) Cumulative mass release of dexamethasone from: PLGA nanoscale fibers (black squares), PEDOT-coated PLGA nanoscale fibers (red circles) without electrical stimulation, and PEDOT-coated PLGA nanoscale fibers with electrical stimulation of 1 V applied at the five specific times indicated by the circled data points (blue triangles). H) UV absorption of dexamethasone- loaded PEDOT nanotubes after 16 h (black), 87 h (red), 160 h (blue), and 730 h (green). The UV spectra of dexamethasone have peaks at a wavelength of 237 nm. Data are shown with a ± standard deviation (n = 15 for each case).(Source: University of Michigan; Re-printed with permission of WILEY-VCH Verlag GmbH & Co. KGaA) | |
| Professor Martin's article, titled "Conducting-Polymer Nanotubes for Controlled Drug Release" was published in the Jan. 25, 2006 online edition of Avanced Materials. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
