| Posted: Oct 24, 2007 | |
First of a kind real-time study of nanosilver in fish embryos raises hopes and concerns |
|
| (Nanowerk Spotlight) What do humans have in common with the pinky-sized tropical zebrafish that zip around in many hobbyists' home aquariums? Well, surprising as it may be, quite a lot actually. Zebrafish share the same set of genes as humans and have similar drug target sites for treating human diseases. For this reason, scientists, when turning to a model-organism to help answer genetic questions that cannot be easily addressed in humans, often chose the zebrafish (Danio rerio) - and save a few mice in the process. Zebrafish are small, easy to maintain, and well-suited for whole animal studies. Furthermore, its early embryonic development is completed rapidly within five days with well-characterized developmental stages. The embryos are transparent and develop outside of their mothers, permitting direct visual detection of pathological embryonic death, mal-development phenotypes, and study of real-time transport and effects of nanoparticles in vivo. Therefore, zebrafish embryos offer a unique opportunity to investigate the effects of nanoparticles upon intact cellular systems that communicate with each other to orchestrate the events of early embryonic development. In a new study, researchers explore the potential of nanoparticles as in vivo imaging and therapeutic agents and develop an effective and inexpensive in vivo zebrafish model system to screen biocompatibility and toxicity of nanomaterials. Such real-time studies of the transport and biocompatibility of single nanoparticles in the early development of embryos will provide new insights into molecular transport mechanisms and the structure of developing embryos at nanometer spatial resolution in vivo, as well as assessing the biocompatibility of single-nanoparticle probes in vivo. | |
| The interest in exploring the use of noble metal nanoparticles – such as gold, silver and their alloys – for diagnostic and therapeutic imaging stems from the drawbacks of current in vivo probes. Fluorescent probes, such as fluorescent dyes and proteins, are not photostable and therefore are useful only for a limited time during the probing event. Quantum dots, on the orther hand, have unique optical properties in comparison with fluorescence probes, such as a tunable narrow emission spectrum, broad excitation spectrum, high photostability, and long fluorescence lifetime. However, they still suffer a certain degree of photodecomposition,it remains a challenge to prevent aggregation of intracellular quantum dots, and they often are extremely toxic. | |
| "The primary challenge of using nanoparticles as probes for in vivo imaging and investigating the effects of nanoparticles on living organisms is (a) to maintain the stability (prevent aggregation and photodecomposition) of nanoparticles in physiological medium and in vivo, (b) to develop real-time imaging tools for tracking the diffusion and location of individual nanoparticles in vivo, and (c) to invent a powerful means to characterize individual nanoparticles in vivo and in real time" Dr. Xiaohong Nancy Xu explains to Nanowerk. | |
| Xu, an Associate Professor in Chemistry and Biochemistry in the Department of Chemistry & Biochemistry at Old Dominion University in Norfolk, Virginia, and her group have accomplished all of these objectives, achieving a major advance in the study of single nanoparticles in vivo. Their study also represents the first rigorous study and characterization of nanotoxicity and nanobiocompatibility ever performed by investigating the effect of highly purified nanoparticles in vivo in real time and by considering the effect of possible trace chemicals from nanoparticle synthesis. | |
| "One can now use the tools (e.g., nanoparticle probes and imaging systems) developed in our study for in vivo imaging and for real-time monitoring the biocompatibility of nanoparticles in vivo" says Xu. | |
| This study has been published in a recent (free access) paper in ACS Nano ("In Vivo Imaging of Transport and Biocompatibility of Single Silver Nanoparticles in Early Development of Zebrafish Embryos"). | |
| With this work, scientists have demonstrated, for the first time, the use of zebrafish embryo as an effective and inexpensive in vivo model system for screening biocompatibility, pharmacological efficacy, and toxicity of nanoparticles. This is a major step forward in efforts to uses nanoparticles as probes to address fundamental questions in biology, and to assess the potential impacts of nanomaterials on environment and human heath. | |
 |
|
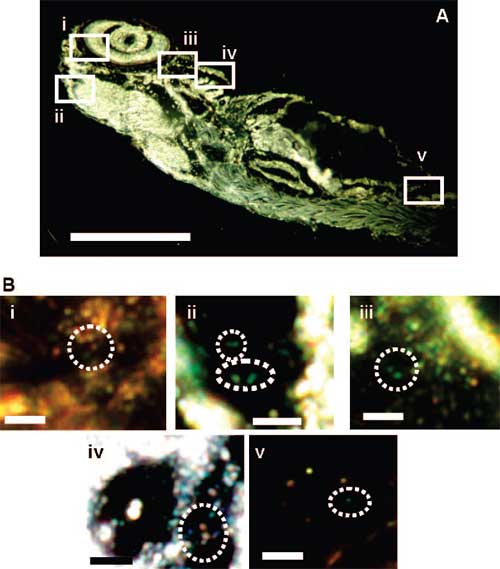
| Characterization of individual silver nanoparticles embedded inside a fully developed (120 hpf) zebrafish using dark-field SNOMS. (A) Optical image of a fixed, normally developed zebrafish. The rectangles highlight representative areas: (i) retina, (ii) brain (mesencephalon cavity), (iii) heart, (iv) gill arches, and (v) tail. (B) Zoom-in optical images of single silver nanoparticles embedded in those tissue sections outlined in (A). Dashed circles outline the representative embedded individual silver nanoparticles. Scale bar = 400 µm (A) and 4 µm (B). (Copyright: American Chemical Society) | |
| "Our study also represents the first rigorous study and characterization of nanotoxicity and nanobiocompatibility ever performed by investigating the effect of highly purified nanoparticles in vivo in real time" says Xu. "This new approach will help set higher standards for the design and evaluation of biocompatibility of nanoparticle probes for in vivo imaging and the characterization of the toxicity of nanomaterials." | |
| Xu explains that they used an optical microscopy technique developed in her group (SNOMS – dark-field single nanoparticle optical microscopy and spectroscopy) to characterize nanometer sizes of individual nanoparticles, based upon inherent brightness and color of individual silver nanoparticles, and to trace individual nanoparticles as they were transported through chorion pore canals into the embryo.? | |
| The nanoparticles show concentration-dependent toxicity – as nanoparticle concentration increases, the number of normally developed zebrafish decreases, while the number of dead zebrafish increases. At lower concentrations (<0.19 nM) the nanoparticles appear to be biocompatible (not toxic). As nanoparticle concentration increases beyond 0.19 nM, only dead and deformed zebrafish are observed, showing a critical concentration of silver nanoparticles in the development of zebrafish embryos. | |
| Apart from the – not unexpected – toxicity findings, the two primary scientific findings of Xu's study are: Single silver nanoparticles resist photodecomposition and blinking and can be directly imaged in embryos for an extended period of time; and individual silver nanoparticles exhibit size-dependent localized surface plasmon resonance spectrum (color). | |
| "These unique features of single nanoparticle optics allow us to 1) distinguish silver nanoparticles from tissue debris and vesicles in embryos; 2) directly image and characterize the sizes of individual nanoparticles in living embryos in real time; 3) probe the diffusion, transport mechanism, and biocompatibility of nanoparticles in living embryos in real time; and 4) investigate the embryonic fluids (e.g., viscosity) at nanometer spatial resolution in real time" says Xu. | |
| The researchers also found that individual silver nanoparticles can passively diffuse into developing embryos via chorion pore canals, create specific effects on embryonic development, and selectively generate particular phenotypes in a dose-dependent manner. Furthermore, the early embryos are highly sensitive to the nanoparticles, showing the possibility of using zebrafish embryos as an in vivo assay to screen the biocompatibility and toxicity of nanomaterials. | |
| This study also raises environmental concerns. It represents the first direct observation of passive diffusion of nanoparticles into an in vivo system, suggesting that the release of large amounts of silver nanoparticles into aquatic ecosystems (e.g., rivers) may have drastic environmental consequences should the sizes of nanomaterials remain unchanged during environmental transport. | |
| Xu points out that her and other group's studies show that it is crucial to further fundamental research to address why some nanomaterials are biocompatible, why some are toxic, and why some may have therapeutic effects. Such studies will lead to rational design of biocompatible nanomaterials for in vivo imaging, diagnosis and therapy. To do this, new tools, such as new assays and new imaging systems, will be needed. | |
| "It will be essential to develop more effective tools for characterization of nanomaterials in situ, and to achieve a better understanding of surface chemistry of nanomaterials and their interaction with biomolecules in vivo" she says. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
