| Posted: Sep 03, 2013 | |
Detecting neurotransmitters in live brain tissue with carbon nanotube electrodes |
|
| (Nanowerk Spotlight) In the brain, one neuron communicates with another by releasing chemicals called neurotransmitters into the 20-40 nm wide gap between them (the synapse). Identifying the chemical nature of these neurotransmitters often is a difficult task and there are many electroactive analytes in the brain that are currently undetectable with conventional technologies due to their low concentrations in the extracellular fluid. | |
| Conventional carbon-fiber electrodes have been the material of choice for in vivo work with fast-scan cyclic voltammetry for the past several decades and they have had a tremendous impact on the field. Unfortunately, they have some limitations that leave some of the molecules that researchers are interested in just out of our reach. | |
| "Though the surface of carbon fibers have been modified in different ways with deposition of various polymers or pretreatments, the underlying electrode substrate has remained the standard carbon fiber," Leslie A. Sombers, an Assistant Professor in the Department of Chemistry at North Carolina State University, explains to Nanowerk. "We feel that further miniaturization of biologically compatible, carbon based electrode materials to the nanoscale promises to enhance the very characteristics that made microelectrodes so transformative in the first place, enabling high speed measurements in discrete spatial locations." | |
| Most experiments that use fast-scan cyclic voltammetry in vivo utilize cylindrical carbon-fiber microelectrodes that are about 100 microns long and 7 microns in diameter. Sombers and her team set out to find a way to improve every aspect of these electrodes used for electrochemical measurements in vivo, to afford them with improved sensitivity, selectivity, and spatial resolution. | |
| "We felt that a disk electrode would be able to sample from smaller locations, improving spatial resolution," says Sombers. "However, reducing the surface area of the electrode – in going from a cylinder to a disk – also reduces its sensitivity. By replacing the carbon-fiber with a carbon nanotube yarn, we have been successful at using a smaller disk electrode in live tissue." | |
| As they report in the August 13, 2013, online edition of ACS Nano ("Carbon Nanotube Yarn Electrodes for Enhanced Detection of Neurotransmitter Dynamics in Live Brain Tissue"), the resulting multiwalled carbon nanotube yarn disk-shaped (CNTy-D) electrodes not only exhibit an improved sensitivity, but also better selectivity because sharper peaks are evident in the voltammograms collected with CNTy-Ds. | |
 |
|
| Scanning electron micrographs of a CNTy-D electrode. (A) Spun MWNTs form a continuous yarn. (B) A single CNTy-D electrode. (C) Detailed image of CNT surface and glass seal of the CNTy-D electrode. (Reprinted with permission from American Chemical Society) | |
| "Furthermore" as Andy Schmidt, a graduate student in Sombers' group, and first author of the paper, notes, "our CNTy-D microelectrodes are customizable and can be tailored to the kinetic and spatial constraints associated with a given experiment." | |
| According to the team, an added – but no less important – benefit is that these microelectrodes also significantly increase the resolution of the voltammograms by providing sharper peaks. This is certainly useful in analyte identification – a critical issue when working in a complex environment such as the brain. | |
| To make the electrodes, the researchers first spun a one-meter long carbon nanotube yarn, with a diameter of approximately 30 µm, from a forest of 40-50 nm diameter individual ultralong multiwalled carbon nanotubes. From this they fabricated microdisk electrodes with an elliptical sensing surface where the ends of the CNTs are still distinguishable. | |
| To experimentally demonstrate that CNTy-D electrodes provide an attractive alternative to standard cylindrical carbon-fiber microelectrodes for work in brain tissue, the scientists inserted single CNTy-D microelectrodes at least 100 µm into a section of a rat brain slice that encompassed the striatum, a region with nerve terminals containing the neurotransmitter dopamine. Then they placed a bipolar stimulating electrode in the vicinity of the working electrode. When they evoked dopamine release with an electrical stimulus, the CNTy-D microelectrode was able to detect the dopamine. | |
 |
|
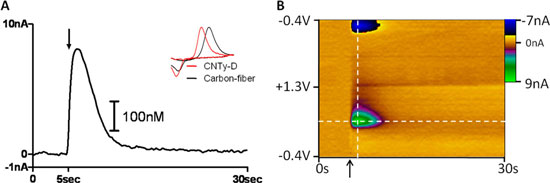
| Endogenous dopamine detection using disk-shaped carbon nanotube yarn electrodes in living brain tissue. (A) Representative current versus time trace collected at the peak oxidation potential for dopamine with concentration determined by electrode calibration. Electrical stimulation occurred at the time indicated by the arrow. Inset: Normalized representative voltammograms corresponding to dopamine on CNTy-D (red) and conventional carbon-fiber (black) electrodes. (B) Three-dimensional color plot of raw data collected in striatal tissue using a CNTy-D electrode. The ordinate is the applied potential, the abscissa is time (s), and the current (nA) is depicted in false color. (Reprinted with permission from American Chemical Society) | |
| The scientists point out that any electrochemical experiment that currently uses a carbon-fiber microelectrodes can benefit from this approach, both in vivo and ex vivo, in tissue and at single cells. | |
| "We hope that future improvements to the new electrodes will enable electrochemical measurements of some of the molecules that currently are hard or impossible to detect with conventional carbon fiber electrodes," says Sombers. "Furthermore, our smaller electrode geometry allows for sampling from smaller environments and detailed investigation of chemical heterogeneity in the brain, eliminating some of the spatial averaging that is inherent to measurements done using cylindrical electrodes." | |
| The team is already working on further miniaturization of their electrode design. By incorporating smaller nanotubes and twisting them more tightly, they hope to reduce the diameter up to five-fold without sacrificing any of the gained sensitivity. They also are investigating functionalization as a means to further improve the detection capabilities. Finally, the roughness of the nanotube yarn surface lends itself well toward the production of enzyme-modified electrodes. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
