| Posted: Mar 31, 2014 | |
Digital nanofluidics - manipulating water droplets on surfaces |
|
| (Nanowerk Spotlight) Within the field of nanofluidics, the manipulation of liquids typically deals with volumes of femtoliters (10-15 L). A femtoliter is one quadrillionth of a liter or 1 µm3. To put that in perspective: that is less than one percent of the volume of an average human cell. | |
| Existing nanofluidic approaches to facilitate the manipulation of ultra-small amounts of liquids usually require their confinement within quasi-1D nanochannels or nanopores. In these devices, the movement of the liquid objects must follow pre-designed routes. | |
| Researchers have now demonstrated a new platform for digital nanofluidics where water nanodroplets are trapped between a mica surface and graphene. Here, with the assistance of a graphene protection layer and ice-like lubricant monolayer, water nanodroplets can be moved, merged, separated, and patterned into regular arrays freely within a two-dimensional channel. | |
| "This is the first demonstration of manipulating individual water nanodroplets of such low volumes on surfaces," Guangyu Zhang, a professor at the Beijing National Laboratory for Condensed Matter Physics and Institute of Physics, Chinese Academy of Sciences, in Beijing, tells Nanowerk. "While our strategy demonstrates the extension of the manipulation freedom from 1D to 2D, we were also able to move water nanodroplets one by one, which means this nanofluidic process is 'digital'." | |
| The work, reported in the March 19, 2014 online edition of ACS Nano ("A Route toward Digital Manipulation of Water Nanodroplets on Surfaces"), also demonstrates the ability to manipulate water nanodroplets with a volume down to the yoctoliter scale (10-24 L). Such small amount of liquid is of great importance not only on study of fundamental physics or chemistry related to its size confinement effect but also for various functional applications. | |
 |
|
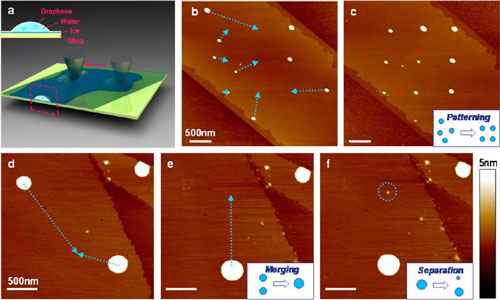
| Manipulation of water nanodroplets (WNs). (a) Schematic for the manipulation of a WN by an AFM tip. Inset shows the sandwich structure of graphene/WN/mica. (b and c) With a multistep translation, the disordered nanodroplets are rearranged into an ordered 3x3 array. The blue dotted arrow (the same below) represents the path of the tip we preset. (d and e) Two nanodroplets are moved to the same location and merged. (f) An ultrasmall nanodroplet with volume as small as 1.2 yoctoliter is separated from a big one in (e). Inset images of (c), (e), and (f) are the sketches for corresponding processes of the manipulation. (Reprinted with permission from American Chemical Society) (click image to enlarge) | |
| Generally, water nanodroplets are hard to form stably on certain surfaces. They are likely to evaporate unless being frozen or under moisture. Thus it's difficult to manipulate them. | |
| "Intrigued by the recent discovery of 2D ice formation between graphene and mica surface by James R. Health’s group at Caltech ("Graphene Visualizes the First Water Adlayers on Mica at Ambient Conditions"), we started to use this mica-water-graphene sandwich structure to realize the water nanodroplets manipulation," Zhang explains the motivation for this work. | |
| Specifically, the researchers put water nanodroplets on a mica surface and covered them with graphene to stabilize them. The one-atom thick graphene membrane works as a protection layer; meanwhile it is also very flexible. | |
| The team found that an ice-like 2D buffer layer formed between the water nanodroplets and the mica surface, acting as a lubricant for the droplets. This made them very mobile and allowed to move them around freely upon applying an external force. The researchers used an atomic force microscope (AFM) to facilitate this manipulation, including moving, merging and separating of these individual water nanodroplets. | |
| Zhang points out that the volume of the smallest manipulable water nanodroplet in their system is around yoctoliter scale, which is a more than 5 orders of magnitude improvement over the existing micro/nanofluidic manipulation limits. | |
| Practically, the demonstrated nanofluidic platform provides a relatively simple solution for lab-on-a-chip applications on which one could carry out molecular analysis, chemical reactions, as well as microelectronic and bioengineering applications. | |
| "The inertia and impermeability of graphene makes it suitable for bearing many kinds of chemical solvent or biological molecules," says Zhang. "And since the typical volume of liquid in our samples is ∼1-100x10-24 L, the synthesis and analysis processes could be low-cost, fast response and environmentally friendly." | |
| Going forward, the team will extend the current, relatively simple platform to a more complex one with liquids other than water. They are also planning to carry out prototype demonstrations of certain functionalities of this platform. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
