| Posted: Apr 09, 2014 | |
A novel approach to designing drug-loaded nanoconstructs |
|
| (Nanowerk Spotlight) Cancer researchers are looking to nanoparticles as a drug carrier capable of localizing and directly releasing drugs into the cell nucleus, thereby circumventing the multi drug-resistance and intracellular drug-resistance mechanisms to effectively deliver drugs to the vicinity of DNA, leading to a high therapeutic efficacy. | |
| Nanomaterials for nanomedicine and biological applications are often two-component structures – referred to as 'nanoconstructs' – consisting of a 'hard' nanoparticle core and a 'soft' shell of biomolecular ligands. In a previous Nanowerk Spotlight we reported on how researchers were able to directly observe how nanoconstructs trafficked to the nucleus via a shuttling protein produced invaginations in the nuclear envelope at the site of the drug-loaded nanoparticles ("Direct observation of how nanoparticles interact with the nucleus of a cancer cell"). | |
| Researchers have now demonstrated a nanoconstruct with enhanced in vitro efficacy. Reporting their findings in the April 1, 2014 online edition of Nano Letters ("Improved in Vitro Efficacy of Gold Nanoconstructs by Increased Loading of G-quadruplex Aptamer"), a team led by Teri W. Odom, Board of Lady Managers of the Columbian Exposition Professor of Chemistry and Professor of Materials Science and Engineering at Northwestern University, reports how oligonucleotides with secondary structure—DNA aptamers with a quadruplex structure—can be highly loaded on the surface of gold nanostars (AuNS) using a citrate buffer at low pH. | |
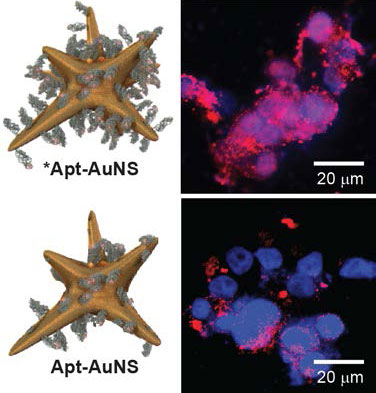
| "Our highly loaded nanoconstructs are special since the gold nanoparticle core has an anisotropic shape and the DNA aptamer we use is an anti-cancer drug," Odom tells Nanowerk. "This highly loaded nanoconstruct (*Apt- AuNS) was taken up by pancreatic cancer cells and fibrosarcoma cells at faster rates and with ca. twice the number compared to Apt-AuNS." | |
 |
|
| Highly loaded nanoconstructs (*Apt-AuNS) can internalize in cancer cells at much faster rates compared to Apt-AuNS with lower aptamer drug loading. (Image: Odom research group, Northwestern University) | |
| The Northwestern team found that the increased loading of Apt on AuNS also resulted in an enhanced in vitro response. | |
| "Moreover" Odom says, "when similar quantities of AuNS were internalized by cancer cells, but having different densities of Apt, we found that *Apt-AuNS showed on average a 42% increase in cancer cell death compared to Apt-AuNS." | |
| She explains that the ligand shell on nanoconstructs with gold nanoparticle cores is usually assembled via a 'salt-aging' process, which requires excessive amount of oligonucleotides and two days to complete. "Compared to this conventional technique, our approach using a citrate-buffer method has three advantages: 1) the amount of initial DNA aptamer needed is reduced significantly; 2) the assembly time takes only three hours; and 3) higher loading of aptamer was possible—2.5 times that compared to salt-aging." | |
| Besides these key, practical advantages, there are two important scientific results from *Apt-AuNS: 1) nanoconstructs with higher amounts of aptamer drug were taken up by cancer cells more rapidly; and 2) higher loading of drug molecules on the AuNS core produced greater cancer cell death. | |
| Hence, increasing the loading density of therapeutic oligonucleotides on nanoparticles could provide a simple means to improve the efficacy of nanoconstructs in cancer treatment. | |
| These results show that high densities of oligonucleotides having secondary structures can now be loaded onto gold nanoparticles—and their therapeutic abilities are maintained. | |
| Another implication is that simply increasing the density of therapeutic oligonucleotides on nanoparticles can significantly improve anti-cancer effects. | |
| Finally, as Odom points out, the importance of the local nanoscale ordering of molecules on nanoparticles has not be heavily pursued for biological applications – but it can offer a different and more efficacious approach for designing nanoconstructs. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
