| Posted: Dec 06, 2007 | |
Nanobioelectronic system triggers enzyme activity |
|
| (Nanowerk Spotlight) Molecular bioelectronics is a rapidly progressing research field that is concerned with the movement of electrons and ions within living systems. This field brings together the strengths of biological reactions and biochemical interactions from nature with electronic signal detection and amplification of human electronics technology. The result will be new applications in medicine, diagnostics and therapeutics that would never be imaginable within the limitations of each separated domain. | |
| One day, bioelectronic devices could even be be directly involved in electrical communication with signal producing or controlling molecules, organelles or structures within our bodies. A more short-term goal, though, is the development of biosensors (such as enzyme-based sensors and DNA sensors) to combine the extraordinary specificity of biochemical receptors with general purpose microelectronics to develop selective probes for diagnostics, drug screening, and toxin detection. | |
| An example of recent advances in the field is the work of researchers in the U.S. who have designed an integrated nanobioelectronic system, exploiting the distinct properties of nanowires and carbon-nanotubes (CNTs), for triggering reversibly and on-demand bioelectrocatalytic transformations of alcohols. Such an adaptive magneto-switchable nanobioelectronic system exploits the distinct properties of its individual nanomaterial components, can be extended to a wide range of biocatalytic systems, and offers great promise for regulating the operation of biofuel cells, bioreactors, and biosensing devices in response to specific needs. | |
| The control of electrocatalytic and bioelectrocatalytic transformations through magnets has been demonstrated several years ago. Here the attraction and retraction of functionalized magnetic spheres is used for ON–OFF switching of enzymatic reactions ("Magnetic Control of Electrocatalytic and Bioelectrocatalytic Processes"). | |
| "Here we report on an adaptive nanomaterial bioelectronic system, integrating nanowires and carbon-nanotubes, for on-demand bioelectrocatalytic transformations" Dr. Joseph Wang says about his group's recent paper i��Chemical Communications ("Adaptive nanowire–nanotube bioelectronic system for on-demand bioelectrocatalytic transformations"). Wang is Director of the Center for Bioelectronics and Biosensors, and a Professor of the department of Chemical Engineering, and department of Chemistry and Biochemistry at Arizona State University. | |
| "The integrated nanowire–CNT system relies on nickel-gold nanowires, which have an enzyme trapped halfway along them, and a CNT-modified amperometric transducer" Wang explains. "Switching the orientation of the nanowires from vertical to horizontal – done by a magnetic field – brings the enzyme in contact with the CNT coated electrode, which allows the enzyme - in this case alcohol dehydrogenase (ADH) - to catalyze the transformation of ethanol to acetaldehyde." | |
| Regeneration of the enzyme's cofactor (NAD+) by the nanotube surface maintains the catalytic activity, and allows analysis by electrochemical methods. | |
| When the nanowires are switched to the vertical position, the reaction is inhibited because the enzyme has no contact with the electrode. Apart from its usefulness as an alcohol sensor, this switchability could hold great promise for regulating the operation of biofuel cells or bioreactors. | |
 |
|
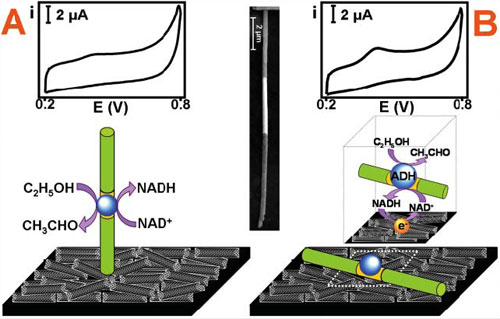
| On-demand magnetically-triggered bioelectrocatalytic transformations of ethanol. The nanowire–CNT based activation of ADH involves Ni/ADH–Au/Ni nanowires in connection to a CNT-coated surface and changes in the nanowire orientation between the vertical (A) and horizontal (B) positions for blocking and activating the bioelectrocatalytic process, respectively. Insets show the corresponding cyclic voltammograms. Also shown (center) is an SEM image of the three-segment nanowire. (Reprinted with permission from RSC Publishing) | |
| The researchers explain that it is also possible to functionalize the adaptive nanowires with two enzymes for the parallel sensing of two alcohols. In this case, the selective analysis of the two alcohols can be accomplished by monitoring the CNT-promoted electrocatalytic signals of their NADH and hydrogen peroxide products during the horizontal alignment of the nanowires. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
