| Posted: Jun 09, 2014 | |
Novel nanocarbon architecture makes a superior bifunctional electrocatalyst |
|
| (Nanowerk Spotlight) Renewable and high-capacity energy systems like fuel cells and metal-air batteries are key components in any scenario on future energy systems free of fossil fuels. | |
| The key electrode reactions for such energy systems are multi-electron processes called oxygen reduction reaction (ORR) and oxygen evolution reaction (OER). Since the sluggish kinetics of ORR is the major factor impeding large-scale application of fuel cells, most research focuses on developing efficient catalysts for ORR and, to a lesser degree, on OER. | |
| The performance of fuel cells largely depends on the ORR – the process that breaks the bonds of the oxygen molecules – which is substantially affected by the activity of the cathode catalyst. Platinum is a very efficient catalyst for boosting the ORR rate. However, it has only moderate activity for the OER, which is the anode reaction and plays an important role in energy conversion and storage. | |
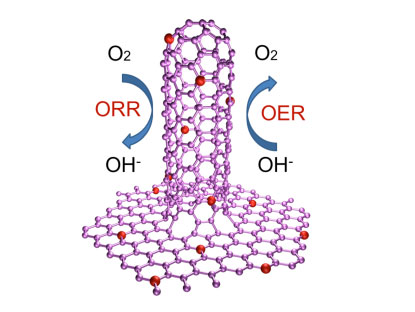
| Researchers from Tsinghua University in Beijing, led by professors Qiang Zhang and Fei Wei, have now demonstrated the synthesis of a novel N-doped graphene/single-walled carbon nanotube (SWCNT) hybrid (NGSH) material by a facile and cost-favorable one-step CVD method. The team reported their findings in a recent edition of Small ("Nitrogen-Doped Graphene/Carbon Nanotube Hybrids: In Situ Formation on Bifunctional Catalysts and Their Superior Electrocatalytic Activity for Oxygen Evolution/Reduction Reaction"). The work is also featured on the cover of the June 2014 print issue. | |
 |
|
| One step growth of nitrogen doped graphene/carbon nanotube hybrids for oxygen reduction and oxygen evolution reaction. (Image: Department of Chemical Engineering, Tsinghua University) | |
| The team's findings open up new avenues for energy conversion technologies based on earth-abundant, scalable, noble-metal-free catalysts. | |
| "In spite of high catalytic activity, the conventional noble metal catalysts like platinum, ruthenium, and iridium all suffer from their high cost and poor stability," Zhang, an associate professor at the Department of Chemical Engineering, explains to Nanowerk. "As a result, scientists are seeking for substitute catalysts from non-noble metal and even non-metal materials. Heteroatoms doped nanocarbon materials afford much improved reactivity and catalytic performance. Our group explored the in situ growth of N-doped graphene and single-walled carbon nanotube hybrids and their superior electrocatalytic performance for ORR and OER." | |
| "We employed the layered double hydroxides as the bifunctional catalyst for the simultaneous growth of graphene and SWCNTs, forming the three dimensional interconnected network," adds Wei. | |
| Zhang's and Wei's groups already have performed a lot of research on the synthesis of hierarchical nanocarbon materials with layered double hydroxides as the catalysts and achieved great progress with many outstanding papers published. | |
| "As for the two typical nanocarbon materials, one-dimensional CNTs and two-dimensional graphene sheets, both of them are inclined to aggregate or stack with each other due to the strong van der Waals forces," Zhang points out. "That prevents the full utilization of the active sites for catalytic reactions. In fact, the integration of graphene and CNTs into a hybrid material is quite a promising strategy to enhance the dispersion of graphene and CNTs, to inherit the advantages of both graphene and CNTs, and to obtain an efficient and effective electronic and thermal conductive 3D network." | |
| For their novel CNT/graphene hybrid material, the researchers employed layered double hydroxides (LDHs) – mainly MgO and Mg2Al2O4 – as the bifunctional catalyst precursors for NGSH formation. Iron-containing nanoparticles were embedded on the LDH plates to tune and control the growth of the CNTs. | |
| As the researchers point out, the FeMoMgAl LDHs derived bifunctional catalysts embedded with thermally stable Fe nanoparticles not only served as an efficient catalyst for the growth of N-doped SWCNTs, but also supplied a lamellar substrate for the templated deposition of N-doped graphene – "therefore, the simultaneous growth of N-doped graphene and SWCNTs can be achieved with the covalent C-C bonding connection." | |
| Based on this concept, Gui-Li Tian, a graduate student and first author of the paper, developed an in situ chemical vapor deposition strategy for the graphene/SWCNT hybrid synthesis. | |
| "N-doped graphene and SWCNTs are intrinsically dispersed in this novel carbon architecture and the N-containing functional groups well dispersed in the conductive scaffold,"says Tian. "The as-fabricated hybrids are with a large surface area, high porosity and also high graphitic degree. All these characteristics render the N-doped graphene/SWCNT hybrids with a high ORR activity, much superior to the two constituent components individually and even comparable to the commercial 20 wt% Pt/C catalysts – but with much better durability and resistance to crossover effect." | |
| As icing on the cake, the team demonstrated that this novel nanocarbon architecture is also electrocatalytically active for OER. | |
| "Our findings indicate that the hybrid material is a promising bifunctional electrocatalyst for regenerative fuel cells and rechargeable metal-air batteries involving oxygen electrochemistry," says Dr. Dingshan Yu from Nanyang Technological University, Singapore, a co-author of the paper. | |
| "We expect that, compared with random graphene and CNTs, more potential applications may arise if the enhanced electrical and optical properties of doped graphene/CNT hybrids are fully exploited," notes Zhang. | |
| The researchers point out that this work also provides a structural platform toward the design of three-dimensional (3D) interconnect materials with extraordinary electron pathways as well as tunable surface/interface that can be used in areas, such as catalysis, separation, drug delivery, energy conversion and storage. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
