| Posted: Jul 28, 2014 | |
Questions arise on the stability of graphene as a platform for photocatalysis |
|
| (Nanowerk Spotlight) Ever since its discovery in 2004, graphene has been considered a relatively stable, high surface area platform to anchor nanostructured catalyst materials for various electrochemical and photocatalytic applications. The emergence of solution-based graphene in the form of graphene oxide has enabled new wet-chemistry approaches to the creation of graphene-based nanocomposites. | |
| These water-dispersible graphene oxide (GO) and reduced graphene oxide (RGO) materials have been extensively investigated as conductive supports in photocatalysis, fuel cells and batteries (see for instance: "Multifunctional graphene mats for sensor and catalyst applications"). | |
| "Basically all studies in this field have considered graphene oxide as a stable platform for electrocatalysis and photocatalysis," Prashant V. Kamat, the John A. Zahm Professor of Science in the Department of Chemistry & Biochemistry and Radiation Laboratory at the University of Notre Dame, tells Nanowerk. "No one has looked into long-term stability and pointed out the susceptibility of these 2D materials to oxidative attack." | |
| A new study coming out of Kamat's lab raises questions about the long-term stability of RGO in an aqueous environment where hydroxyl radicals can be present as part of the photocatalytic reaction cycle. | |
| The motivation for this work stems from the photocatalytic activity of TiO2 nanoparticles. Since these semiconductor nanoparticles readily oxidize organic compounds under UV irradiation, the scientists hypothesized that it should also be possible to oxidize graphene oxide with them. | |
| Indeed, they were able to demonstrate the susceptibility of graphene based materials to hydroxyl radicals attack as they undergo fragmentation to produce PAH (polyaromatic hydrocarbons) type species and eventually to complete mineralization. | |
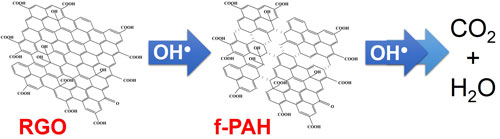 |
|
| Depiction of the oxidative fragmentation and mineralization of RGO during irradiation of TiO2-RGO suspensions. Initial OH• attack leads to scission of the RGO sheets into PAH-like compounds as evident in the rapid decrease in RGO visible absorbance. Continued irradiation promotes the slower mineralization of these fragments to CO2 and H2O. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| As the team reports in the July 21, 2014 online edition of Chemistry of Materials ("Is Graphene a Stable Platform for Photocatalysis? Mineralization of Reduced Graphene Oxide with UV-Irradiated TiO2 Nanoparticles"), the results show a significant weakness in the otherwise robust material when used in certain scenarios where it may undergo oxidative attack by highly reactive oxidizing species. | |
| "While our work focuses specifically on photocatalytically generated hydroxyl radicals, many electrocatalytic systems where graphene has found its use – such as fuel cells – also involve the hydroxyl radical as an undesired player in the overall catalytic reaction scheme," Kamat points out. | |
| According to the researchers, the oxidative fragments formed during initial attack of hydroxyl radicals have the potential to induce toxicity in the environment since they are chemically similar to many common environmental contaminants such as polyaromatic hydrocarbons. | |
| "The oxidative degradation of RGO by UV-irradiated TiO2 fundamentally challenges the current paradigm of coupling these materials for aqueous-based photocatalysis and other applications where hydroxyl radicals are present," Kamat sums up the findings. "In fact, the fragmentation of RGO could exacerbate the environmental challenges TiO2-RGO composites are employed to remediate. Since the initial breakdown of the RGO occurs quickly and leads to more recalcitrant compounds that are slower to degrade via hydroxyl radicals, these fragments, if untreated, may enter into the environment." | |
| The results of this study clearly point out the need for a better understanding of the long-term stability of graphene materials in electro- and photocatalytic applications, particularly those involving hydroxyl radical intermediates. If radical attack leads to degradation, new approaches in designing the electrochemical and photocatalytic assemblies using graphene as a support must be explored. | |
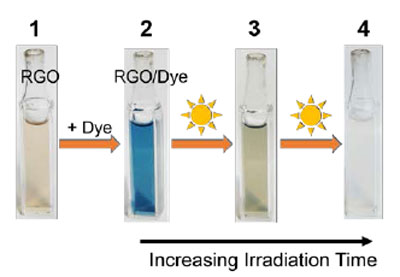 |
|
| Simultaneous photocatalytic degradation of Naphthol blue black dye and RGO using UV/visibe-irradiated TiO2 suspension. Visual depiction of color changes (1) initial RGO suspension, (2) upon addition of Naphthol blue black dye, (3) 10 minutes of UV irradiation, and (4) 100 minutes of UV irradiation, at which point the solution is completely colorless. (Reprinted with permission by American Chemical Society) | |
| Kamat notes that a positive finding of this work is the ability to mineralize graphene-based materials with hydroxyl radicals since it demonstrates a hydroxyl radical-mediated approach to remediation of graphene and potentially other similar nanocarbons such as carbon nanotubes, C60, C70, etc. | |
| As an example, the team carried out photocatalytic degradation of a textile diazo dye (naphthol blue black) in nanoscale TiO2 and nanoscale TiO2/RGO suspensions. Upon exposure to UV irradiation both dye and RGO degrade over time, indicating the successful mineralization process. | |
| Kamat's group will build on these findings by investigating new approaches for making graphene materials more stable in an aqueous environment with the presence of hydroxyl radicals. They also plan to extend this approach to mineralize different nanocarbon materials. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
