| Posted: Aug 14, 2014 | |
Upconverting synthetic leaf takes it cues from nature |
|
| (Nanowerk Spotlight) A large part of low-energy photons, such as in the deep-red and infrared, are lost during conventional photovoltaic or photochemical processes. However, about half of all the solar energy reaching the Earth's surface can be found in these wavelengths. | |
| Harvesting this light more efficiently is possible thanks to a process called photon energy upconversion where two or more weak captured photons are converted into a single strong, energy-rich photon. | |
| So far, triplet-triplet annihilation photon energy upconversion (TTA-UC) is the only upconversion method that has been experimentally demonstrated to operate with noncoherent low intensity illumination such as sunlight ("Up-Conversion Fluorescence: Noncoherent Excitation by Sunlight"). | |
| However, the solid TTA-UC materials reported so far have focused mainly on elastomeric matrices with low barrier properties because the TTA-UC efficiency generally drops significantly in glassy and semicrystalline matrices. | |
| "To fully exploit the TTA-UC process in different applications, the importance of the development of solid-state-like TTA-UC materials for better device integration cannot be neglected since such materials are easily processable," Katharina Landfester, a professor at the Max Planck Institute for Polymer Research (MPI-P) in Mainz, Germany, tells Nanowerk. | |
| In new work, Landfester and her group have successfully synthesized a bioinspired upconverting solid-state-like film using nanocellulose. They reported their findings in the July 14, 2014 online edition of ACS Nano ("Photon Energy Upconverting Nanopaper: A Bioinspired Oxygen Protection Strategy"). | |
| As Landfester explains, the TTA-UC process uses a dense populated sensitizer triplet ensemble, created by optical excitation, as energy reservoir for further energy transfer. In oxygen-rich environment major part of this stored energy is lost during the quenching process stimulated by the molecular oxygen presence. | |
| Therefore, in order to keep a high upconversion efficiency, the molecular system for this process has to be protected against quenching. For their design, the MPI-P researchers followed a bio-inspired strategy by using the same protection material that nature uses in the structural buildup of leaves – cellulose. | |
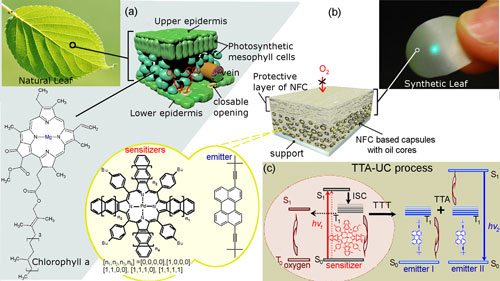 |
|
| (a) Structural buildup of a natural leaf, showing the protective outer epidermis layers and the closable openings through which the flow of CO2 and O2 is regulated. Between the epidermis layers, the liquid-filled parenchyma cells (palisade and spongy mesophyll) are found. The chlorophyll is present in the chloroplasts that are found within the mesophyll cells. (b) Structure of the synthetic leaf showing the (high oxygen barrier) protective layer of cellulose nanofibers and the embedded oil-core capsules underneath. The sensitizers and emitter are present in the liquid cores of the capsules. The cyan dot on the synthetic leaf is the UC emission obtained at broad-band excitation. The synthetic leaf (inset) was decoupled from its underlying support. The scattered excitation radiation is blocked by a short pass filter (SP01-633RU-25, Semrock Inc.). (c) Simplified scheme for the TTA-UC process in multicomponent organic systems and quenching of the TTA-UC process (the brownish circle) in the presence of molecular oxygen. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| By using pristine cellulose nanofibers, the team achieved a 'green' functional nanomaterial with ultrahigh barrier properties. Since the cellulosic nanofibers are sustainable, biodegradable, and derived from an inexhaustible plant source, these results could pave the way for the creation of a fully recyclable and inexpensive TTA-UC solid-state-like film. | |
| The semi-crystalline cellulose used in this work consists of amorphous cellulose – ensuring mechanical flexibility and processability of the mew material, using conventional printing techniques – and crystalline nanorods – ensuring high oxygen barrier properties. | |
| The researchers fabricated their synthetic leaves by covering a layer of nanocellulose-based capsules with a diameter of 1.2 ±0.3 µm and a wall thickness of about 30nm, containing chromophores in the liquid core, with a high oxygen barrier protective layer of cellulose nanofibers. By altering the thickness of this protective layer, the rate of gas transport to the capsules can be regulated. | |
| "Using the same material for the polymer film, delivering distinct mechanical properties, and for the shell of the upconversion nanocontainer, drastically reduces the free volume of the new materials, thus substantially reducing the oxygen permeability," explains Landfester. | |
| She cautions, though, that the analogy with the protection strategy used by nature is incomplete: "Besides low oxygen permeability – the 'passive protection' – photosynthetic plants use a complicated mechanism for chemically deactivating singlet oxygen (created during the quenching process) and various radicals – i.e. 'active protection'. Our next step is to combine low permeability with active scavenging of the created molecular oxygen." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
