| Posted: Oct 29, 2014 | |
A nano-thermodynamic look at gold-copper alloys |
|
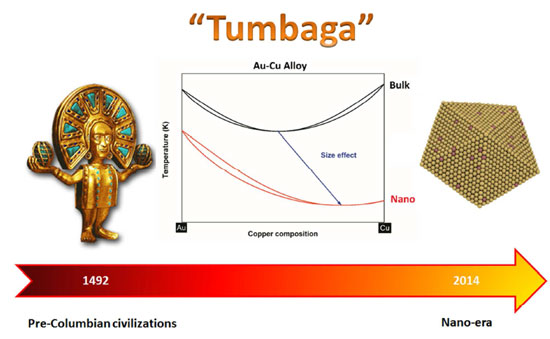
| (Nanowerk Spotlight) The Spaniards called it Tumbaga – an alloy of gold and copper which they found in widespread use by the pre-Columbian cultures in Central America. The popularity of this alloy comes from its congruent melting point, i.e. at a particular composition, the alloy behaves like a pure element (i.e. it melts at a definite temperature rather than over a range) and also the melting point of the alloy is reduced as compared to the two pure elements. For bulk Au-Cu, the congruent melting point occurs at 44% copper composition and 910°C, well below the gold melting point (1064°C) and the copper melting point (1084°C). | |
| Today, gold-copper alloys are very popular catalysts in nanotechnology, for instance to efficiently convert carbon dioxide or to help fabricate a more powerful and longer lasting fuel cell material. | |
| "This alloy exhibits novel physical and chemical properties at the nanoscale," Dr. Grégory Guisbiers from the Department of Physics & Astronomy at the University of Texas at San Antonio (UTSA), tells Nanowerk. "Although the Au-Cu alloy has been extensively studied in the literature both at the bulk and nano-scales, the prediction of phase diagrams at the nanoscale is still missing." | |
 |
|
| (© RSC Publishing) | |
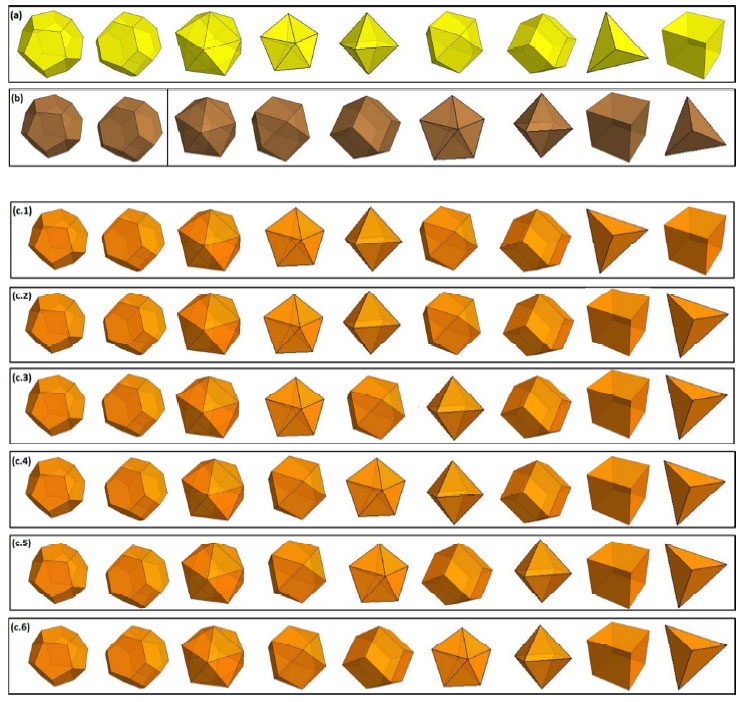
| In a new paper published in the October 22, 2014 online edition of Nano Letters ("The Gold-Copper Nano-Alloy, 'Tumbaga' in the Era of Nano: Phase Diagram and Segregation"), Guisbiers and fellow scientists from UTSA present the phase diagram of Au-Cu at the nanoscale for the relevant distinct polyhedral morphologies of nanoparticles at sizes 4 nm and 10 nm – namely the tetrahedron, cube, octahedron, decahedron, dodecahedron, rhombic dodecahedron, truncated octahedron, cuboctahedron, and icosahedron. | |
| The team from Prof. Miguel Jose Yacaman's group used a thermodynamic approach that gives some interesting insights concerning the behavior of several polyhedral Au-Cu nanoalloys. | |
| "Our results illustrate how the dynamic and structural behavior of the alloy as calculated at the special sizes 10 and 4 nm, evolves from the gold sequence to the copper sequence as the copper composition increases" notes Guisbiers. "Whatever the composition and the size of the nano-alloy, the preferred shapes remain the dodecahedron, truncated octahedron and the icosahedron. Shapes with a Cu-rich core/Au-rich surface are the most stable structures." | |
 |
|
| Predicted sequence of preferred shapes (left to right) for a) gold, b) copper and c) copper-gold alloy at 10nm and 4 nm where 1, 2, 3, 4, 5 and 6 represent different range of composition. (Reprinted with permission by RSC Publishing) (click on image to enlarge) | |
| The paper highlights the size and shape effects on the congruent melting point, showing its copper enrichment when size decreases: For each shape investigated, by decreasing the size, the congruent melting point is shifted to lower temperature and higher copper composition. | |
| Previous work has shown that among bimetallic systems built with elements of the same group in the periodic table, the trend to be in the core is higher for the element with the smaller core electron density (copper in this case) i.e. the element with the higher core electron density (gold in this case) will be located at the surface. The structures predicted thermodynamically in this work are in good agreement with these observations. | |
| "The trend observed from our approach can also be used as a starting point for ab initio density functional theory (DFT) methods to predict the behavior of smaller Au-Cu clusters," concludes Guisbiers. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
