| Posted: Mar 24, 2015 | |
A quick and simple blood test to detect early-stage cancer |
|
| (Nanowerk Spotlight) Early and accurate detection of cancer is critical for successful cancer therapies. In most cases, a tissue biopsy is the initial means of making a diagnosis after some form of cancer screening has come up with a positive result. However, tumor antigen-specific autoantibodies – antibodies produced by the immune system that are directed against one or more of the individual's own proteins – are known to appear months even years before clinical diagnosis of cancer, and autoantibodies have been found in many types of cancer. | |
| With increasing accuracy, nanosensor platforms for detecting cancer biomarkers are becoming viable complements to invasive biopsies of metastatic tumors (although, for the time being, it still appears likely that biopsies will have to be used for a final diagnosis). Unfortunately, despite recent advances in nanotechnology cancer diagnostics, non-invasive screening test for early-stage detection of most cancer types are almost non-existent. | |
| "Most studies are attempting to identify tumor-specific antigens and detect antibodies that are specific to individual tumor-associated antigens," Qun Huo, an Associate Professor in the NanoScience Technology Center at the University of Central Florida, explains to Nanowerk. "Different from these approaches, the novel nanoparticle test that we developed detects an overall increase of human immunoglobulin G (IgG), including the tumor-specific autoantibodies, adsorbed to a gold nanoparticle surface." | |
| "On one hand, this test may not be able to identify the specific type of cancer; on the other hand, this test may potentially be able to detect early stage tumor-induced immune responses associated with a broad spectrum of cancer types, making this test potentially a universal screening test for cancer risk assessment," she points out. | |
| Huo worked as part of an interdisciplinary team that reported their findings in the March 10, 2015 online edition of ACS Applied Materials & Interfaces ("Gold Nanoparticle-Enabled Blood Test for Early Stage Cancer Detection and Risk Assessment"). | |
| The new blood test is extremely simple: it comprises two simple liquid mixing processes. The test results are straightforward, easy to interpret, and do not require any further data processing or statistical analysis. The cost of the test is low: the material cost is less than $1 per test. The test results are obtained within minutes. | |
| There are two innovative elements in this work, and both innovations are based on the unique properties of gold nanoparticles. | |
| The first one is the development of a gold nanoparticle-enabled dynamic light scattering assay (NanoDLSay) technique for molecular detection. | |
| The second one is the discovery of cancer biomarker molecules from the 'protein corona' formed on the gold nanoparticles upon adsorption of blood serum proteins to the nanoparticle surface. | |
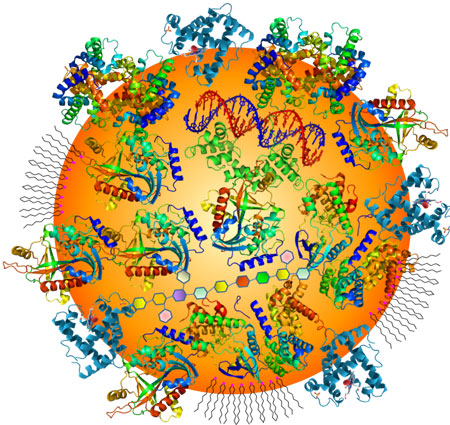 |
|
| Figure 1: Illustration of a protein corona formed on a gold nanoparticle (AuNP) surface upon adsorption of blood serum proteins and other biomolecules. (Image: Dr. Huo, University of Central Florida) | |
| "With these two innovations, we developed a simple blood test that detects molecular differences between cancer patients and healthy controls," says Huo. "Early cancer detection faces two-fold challenges: one is to discover the biomolecules that can become cancer biomarkers, and the second one is the development of suitable techniques for the detection of these cancer biomarker molecules. Our development opens new doors for potential solutions to both challenges." | |
| The new blood test exploits the unique optical property of gold nanoparticles, well known for their strong light scattering at visible light region. Researchers have been using this optical property of gold nanoparticles for biomolecular and cell imaging for a while, because gold nanoparticles can easily stand out from the background scattering of biological cells under the dark-field optical imaging mode. | |
| Dynamic light scattering (DLS) is a technique used routinely for nanoparticle size characterization and analysis. DLS relies on the light scattering property of nanoparticles for size measurement. Most researchers working in the nanoparticle field either have DLS instruments in their own laboratories or have ready access to DLS instruments at their central facility. Moreover, DLS is a relatively inexpensive and easy-to-use instrument. | |
| "Our unique contribution is to combine the strong light scattering property of gold nanoparticle probes with the size measurement capability of DLS, and to create a quantitative analytical technique, nanoparticle-enabled dynamic light scattering (NanoDLSay) for chemical and biomolecular detection and quantitative analysis," notes Huo. | |
| NanoDLSay detects chemical and biological target molecules and species by simply measuring the size change of the gold nanoparticle probes upon binding with target analytes. | |
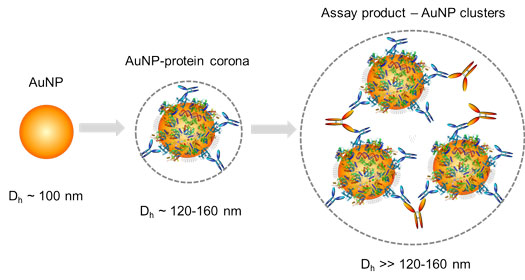 |
|
| Figure 2: Detection of autoantibodies and other target proteins in the gold nanoparticle protein corona by measuring the average particle size (hydrodynamic diameter Dh) change of the nanoparticles upon binding with specific antibodies to the target proteins. (Image: Dr. Huo, University of Central Florida) | |
| The surface chemistry of gold nanoparticles can be designed to contain specific target-recognizing molecular species such as antibodies, DNA probes, carbohydrate binding proteins, metal-chelating chemical ligands. Upon binding with the target analytes, the analytes cause the gold nanoparticle probes to cluster together. As a result, the average particle size of the assay solution is increased, and such size increases can be readily detected by DLS. | |
| "We and other research groups have so far successfully used NanoDLSay for a wide range of target analyte detection and quantitative analysis, including proteins, DNAs, antibodies and antigens, viruses, carbohydrates, toxic metal ions, small molecules, food and environmental toxins," says Huo. "The published studies show that as a quantitative technique, NanoDLSay is highly sensitive and reproducible." | |
| The above-mentioned unique surface chemistry of gold nanoparticles also plays another important role in the way this blood test works. It has been known that when nanoparticles, such as citrate-capped gold nanoparticles are mixed with blood, the proteins and other biomolecules will adsorb to the nanoparticle surface to form a layer of 'protein corona'. Although the concept of protein corona is most frequently used, other non-protein biomolecules such as lipids, DNAs, carbohydrates could also be part of the corona. | |
| The team's contribution is that they first proposed a hypothesis that the molecular composition of this protein corona could reveal the cancer status. | |
| "When a tumor starts to develop in human body, many chemical and biochemical reactions will occur in the tissue and the blood," explains Huo. "Our rationale was that the overall composition and the relative quantity of biomolecules in a cancer patient?s blood should differ from a healthy person, therefore, when a gold nanoparticle is mixed with a blood serum, the type and amount of proteins and other biomolecules adsorbed to the nanoparticle surface could differ between cancer patients and healthy person." | |
| With this hypothesis in mind, the researchers applied their new NanoDLSay technique to study the proteins in the gold nanoparticle protein corona, and indeed, they discovered that a major blood protein, human IgG, is increased in prostate cancer patients. Through proteomic analysis, they also discovered other proteins that may serve as potential cancer biomarkers. | |
| IgG is one of the most abundant proteins in the blood. IgG antibody is a key component of our immune system. Cancer immunology research has revealed that upon tumorigenesis, our immune system will launch defensive actions against the tumor by producing autoantibodies. The team's finding of increased immune activity from prostate cancer patients provided additional evidence on the potential links between cancer and the immune system. | |
| This study is addressing a specific diagnostic problem that exists in the prostate cancer field: to distinguish early stage prostate cancer from non-cancer prostate conditions with an as little as possible error rate. One of the major problems in prostate cancer screening is the over-diagnosis of cancer, because the widely used PSA test has poor specificity in distinguish early stage prostate cancer from BPH (benign prostate hyperplasia). In their study, the scientists found that their new test can distinguish prostate cancer patients from BPH patients with more than 90% specificity. | |
| Whereas the team's analytical technique – NanoDLSay – can be used to address a wide range of biomedical research problems, their current focus is to identify cancer biomarker molecules from gold nanoparticle protein corona, and to develop new blood tests suitable for early stage cancer detection and diagnosis. | |
| "We have so far mainly worked on prostate cancer," says Huo. "We are extending our study to other types of cancer. Our vision is to develop an array of blood tests for early detection and diagnosis of all major cancer types, and these blood tests are all based on the same technique and same procedure." | |
| Going forward, the team's research will focus on two directions: one is continuing new cancer biomarker discovery using the NanoDLSay technique; and the second one is to conduct more extensive clinical studies to validate the new cancer biomarkers discovered in this study. | |
| Dr. Huo co-founded a startup company, Nano Discovery Inc. to commercialize the NanoDLSay technique and the new diagnostic test. Nano Discovery Inc. will present the technology and exhibit their product at the 2015 TechConnect World Innovation Summit & Expo, co-located with the National Innovation Summit and National SBIR/STTR Conference, June 15-17, Washington DC. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
