| Posted: Nov 23, 2015 | |
Janus separator: A new opportunity to improve lithium-sulfur batteries |
|
| (Nanowerk Spotlight) Lithium-sulfur (Li-S) batteries, which employ sulfur as cathode and metallic lithium as anode materials, are emerging as promising alternatives to the widely used lithium-ion batteries because theoretically they can render 3-6 times higher energy density (2600 Wh kg-1). In practice, though, it has proven challenging to approach that theoretical value. In addition, most lithium-sulfur batteries suffer from capacity loss under moderate current densities and self-discharge. | |
| "The two major problems with Li-S batteries stem from the intrinsic inert reaction kinetics of sulfur redox and the unique 'shuttle' mechanism described as that soluble intermediates – polysulfides consisting of lithium-terminated sulfur-chains — diffuse between the cathode and anode, thus being consumed within the battery instead of being utilized," Dr. Qiang Zhang, an associate professor at the Department of Chemical Engineering at Tsinghua University, explains to Nanowerk. "To solve these issues and to improve battery performance relies on not only the electrode materials but also other cell components such as the separator." | |
| The separator – which in secondary batteries and supercapacitors separates the cathode and anode to prevent shorting; while in flow batteries and fuel cells, it should selectively let specific ions pass through while reject others – is a key component in energy storage devices and its unique functionalities are indispensable. | |
| "In a lithium-sulfur battery, the separator should take more responsibility," says Zhang. "In fact as the result of polysulfide shuttle, the discharge products are easily deposited between the separator and the cathode instead of within the cathode. An insulating film is formed on the electrode surface and thereby prevents the further reaction. This might be a negligent reason for low capacity and its fading." | |
| "Previous works tried to retard the diffusion of polysulfides usually by modifying the sulfur cathode," he adds. "But we think the separator is the key to control the shuttle effect and engineered a special separator to improve our lithium-sulfur batteries – and we called it Janus Separator." | |
| Janus is a two-faced god in Roman mythologies and later has been used in science to describe asymmetric structures. A 'Janus structure' can often realize new properties that are inconceivable for homogenous or symmetric structures. | |
| Accordingly, as the scientists at Tsinghua University report in Advanced Science ("Janus Separator of Polypropylene-Supported Cellular Graphene Framework for Sulfur Cathodes with High Utilization in Lithium–Sulfur Batteries"), their Janus Separator is made up of two different layers. The one is a polymeric porous membrane and the other on is a thin film of nanostructured carbon. | |
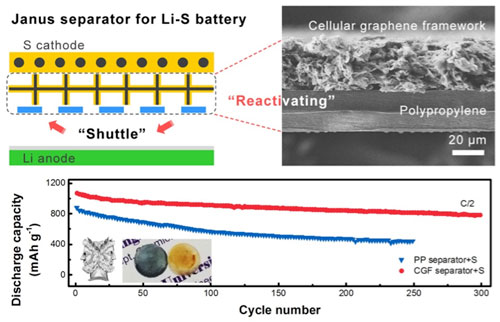 |
|
| Top: Illustration and SEM image of a Janus Separator; Bottom: Cycling performance of Li-S batteries with Janus Separator (red line) and routine separator (blue line). Inset figure is the Janus God and photos of Janus separator and routine separator after use. (Image: Dr. Qiang Zhang, Tsinghua University) (click on image to enlarge) | |
| "The carbon layer we designed for our Janus Separators is composed of a kind of mesoporous but graphitic carbon," notes Hong-Jie Peng, the paper's first author. "It looks like a micrometer-sized honeycomb where many graphene cages are compactly packed together and interconnected. The size of each cage is around 7-8 nm. Compared to previously reported microporous carbon or carbon blacks, this carbon material exhibits much larger pore volume so that it can accommodate more solid products while remains the ion channel." | |
| He points out that the graphitic nature also leads to superior electrical conductivity for fast charge transport. | |
| "Actually the Janus separator outperformed separators made of other carbon materials tested in our experiments," he says. | |
| Dai-Wei Wang, another co-author of the paper, tested the lithium-sulfur battery prototype with a Janus Separator and routine sulfur cathode blended with carbon blacks. Both the capacity and cyclic stability is improved by 40% when compared with that with a routine separator. | |
| "The most exciting result for us is that accompanied with improvements in capacity and stability, the passivated film induced by shuttle phenomenon completely disappear by employing our Janus Separator," he notes. "We are happy that our hypothesis on capacity decay has been unambiguously supported." | |
| In most reported lithium-sulfur cells, the sulfur content in the cathode and the areal loading amount are both very low as a result of adding high amounts of carbon in order to gain higher capacity than based on sulfur only. However, since only sulfur stores the energy, its low content strongly impedes the overall energy density. | |
| "People might argue that by employing our Janus Separator we actually add more carbon to the whole system so that the battery shows a better performance," says Hong-Jie. "But in the lithium-sulfur batteries we fabricated, the areal loading of sulfur is 5.3 mg cm-2, which is 2-10 times higher than in most reports. And the loading of carbon in Janus Separators is only 0.3 mg cm-2. By employing the Janus Separator with such little carbon addition, the areal capacity is improved from 2.6 mAh cm-2 to 5.5 mAh cm-2." | |
| "In the materials and chemistry world, sometimes not too much change can make a really big difference," comments Qiang. | |
| The concept of a Janus Separator by engineering a carbon layer between the polymer membrane and the sulfur cathode might only be a beginning. According to the team, next steps could lead to further device designs towards anode protection or smart management of mass transportation. | |
| "I believe the Janus Separator will open new opportunities for improving the electrochemical energy storage devices (EESDs)," says Qiang. "Not only for lithium-sulfur batteries but also for other EESDs where the redox materials are mobile and under complicated phase change, the Janus Separator will play a significant role. I am looking forward to more and more progress on novel separators and applications." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
