| Posted: Mar 29, 2016 | |
Two new developments for in-situ electrochemical detection |
|
| (Nanowerk Spotlight) Graphene has been widely used to enhance the performance of energy storage devices due to its high conductivity, large surface area and excellent mechanical flexibility. However, it is still unclear how graphene influences the electrochemical performance and reaction mechanisms of electrode materials. | |
| "To explore the intrinsic mechanisms of the electrochemical reactions of porous graphene oxide in situ, the single-nanowire electrochemical probe is an effective tool," Liqiang Mai, a Professor of nano energy materials and devices at Wuhan University of Technology, tells Nanowerk. "Although graphene is usually used as an additive in active materials to improve the electrochemical performance, how graphene influences the electrochemical performance and reaction mechanisms of electrode materials is under dispute." | |
| To address these issues, Mai and his team have explored single-nanowire electrochemical devices to investigate the capacitance, ion diffusion coefficient, and charge storage mechanisms of graphene. They report their findings in the online issue of Nano Letters on February 19, 2016 ("Single-Nanowire Electrochemical Probe Detection for Internally Optimized Mechanism of Porous Graphene in Electrochemical Devices"). | |
 |
|
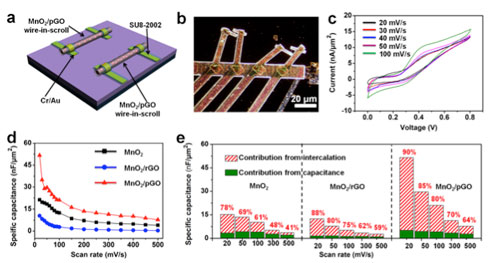
| The schematics illustrations and electrochemical performances of symmetric single nanowire electrochemical devices. (a) Schematic diagram of a symmetric single nanowire electrochemical device. (b) Optical image of a symmetric single nanowire electrochemical device. (c) The CV curves at the scan rates of 20, 30, 40, 50, 100 mV/s for the as-prepared MnO2/pGO symmetric single nanowire electrochemical devices in 6 mol/L KOH. (d) The specific capacitance vs. scan rates of as-prepared symmetric single nanowire electrochemical devices in 6 mol/L KOH. (e) Bar chart showing total capacitance in nF/µm2 together with percentage contribution from capacitive and intercalation as a function of different materials. The values showing here are calculated using CV recorded at the scan rate of 20, 50, 100, 300, 500 mV/s. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| The researchers demonstrate the internally optimized mechanism of porous graphene through these single-nanowire electrochemical devices for the first time: the reduced graphene oxide wrapping around the MnO2 nanowire greatly increases the electronic conductivity of the active materials, but decreases the ion diffusion coefficient because of the shielding effect of graphene. | |
| By creating pores in the graphene, the ion diffusion coefficient could be recovered without degradation of the electron transport rate, which significantly improves the capacitance. | |
| LiFePO4 is one of the most outstanding cathodes for high performance lithium-ion batteries. However, it is limited by its unsatisfactory low temperature performance. | |
| In their work, Professor Mai's group explored the structural dynamics and reaction routes of LiFePO4 via operando condition with high rates, well reproducibility over cycles and low temperature with common laboratory X-ray techniques. | |
| They team reported these findings in the inaugural issue of Nano Energy on February 24, 2016 ("In operando observation of temperature-dependent phase evolution in lithium-incorporation olivine cathode") | |
 |
|
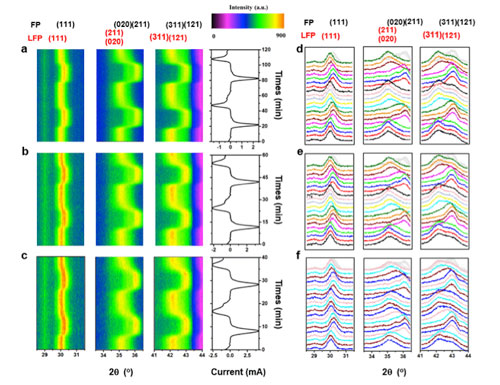
| The capturing of metastable phase at 273 K. (a-c) The image plot of diffraction patterns for the (111), (211), (020), (311), and (121) reflections during the two CV cycles under scan rates of 1.4 (a), 2.8 (b), and 4.2 mV s-1 (c) at a temperature of 273 K. The corresponding current curves are plotted to the right. (d-f) Selected individual diffraction patterns for the two CV cycles at 1.4 (d), 2.8 (e), and 4.2 mV s-1 (f), corresponding to the results in Figure 1a, b and c, respectively at 273 K. (Reprinted with permission by Elsevier). (click on image to enlarge) | |
| The ground-breaking research about the phase transformation routes of LiFePO4/FePO4 at different temperatures (253, 273, 293, and 313 K) are carried by in situ XRD with exceptionally high cyclic voltammetry scan rates of 4.2 mV s-1. | |
| With these results, for the first time, the team demonstrates the existence of intermediate phases during the lithiation/delithiation processes at low temperature. At a temperature of 273 K, the ion diffusion rate along the b direction is greatly limited, which leads to the accumulation of potential. Driven by the overpotential, another channel is forced to open, which results in the stronger polarization and the formation of intermediate phases at lower temperature. | |
| Mai's group also found that such intermediate phases between LiFePO4/FePO4 can efficiently inhibit the degradation of the ion diffusion coefficient with a decrease in reaction temperature. | |
| This indicates that the formation of such intermediate phases or partial amorphism is a potential way to improve the low temperature performance of LiFePO4. | |
| Besides LiFePO4, such a high-time resolution in situ XRD testing method with common laboratory equipment can be used to understand the reaction mechanism of other battery electrode materials and related electrochemical reaction systems. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
