| Posted: Mar 30, 2016 | |
Ionic purity of nanoparticles is key to switching between purification and contamination regimes in liquid crystal devices |
|
| (Nanowerk Spotlight) Modern liquid crystals devices – displays being the most well-known – utilize high resistivity liquid crystals characterized by negligibly small concentration of mobile ions. However, these devices are prone to uncontrolled ionic contamination. | |
| This contamination can easily happen at any stage of the device fabrication or while operating the device. Ions in liquid crystals can compromise the overall performance of the device by leading to many negative side effects such as image sticking, image flickering, and slow response. | |
| Solving these problems requires the development of new methods, suitable for the permanent purification of liquid crystals from ions. | |
| For more than a decade, scientists around the globe have studied the effects of nanoparticles on the electrical properties of liquid crystals. This extensive research was inspired by the very simple idea of using nanodopants as permanent ion capturing agents in liquid crystals. | |
| In other words, nanoscale objects dispersed in liquid crystals can trap mobile ions, thereby reducing their concentration and providing a permanent purification of liquid crystals from ions. | |
| Numerous experimental results (more details can be found in a recently published review1 revealed that the purification regime is not the only possible outcome of the dispersion of nanoparticles in liquid crystals. | |
| While the majority of published results are in favor of the ion trapping scenario (nanoparticles capture ions in liquid crystals), there are some publications reporting opposite effects (nanoparticles release some ions thus causing the contamination of liquid crystals). Moreover, in some special cases, the regime of the contamination can be followed by the regime of the purification. | |
| All these puzzling findings have not been fully explained and became a source of frustration and skepticism among scientists and R&D engineers. The entire approach of purifying liquid crystals from ions by means of nanoparticles was called into question. The key issue in all this is the identification of the major factors that determine whether a technique results in purification or contamination. | |
| The solution to this question has been recently reported in Applied Physics Letters (APL)2. | |
| "By considering nanoparticles as partially contaminated with ions, three possible regimes for liquid crystals doped with nanomaterials can be predicted, namely, the purification, the contamination, and no change at all," Yuriy Garbovskiy, PhD, a researcher at the UCCS BioFrontiers Center & Department of Physics, University of Colorado, explains to Nanowerk. "According to our findings, switching between these regimes is governed by three dominant factors: the purity of liquid crystals, the purity of nanoparticles, and the ratio of the adsorption rate to the desorption rate." | |
| An example of these regimes is shown in the figure below:. | |
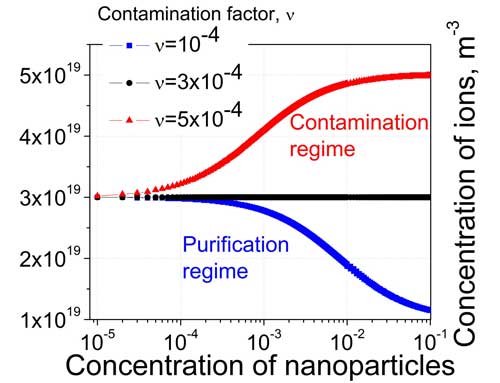 |
|
| The dependence of the concentration of mobile ions in liquid crystals doped with nanoparticles on the weight concentration of nanoparticles. Curves are modeled for nanoparticles characterized by different levels of their ionic contamination factor (the contamination factor of 100% pure nanoparticles is zero). (Image: Dr. Yuriy Garbovskiy, University of Colorado) | |
| Garbovskiy points out that the formalism he developed can explain all, even seemingly contradictory, results reported in the literature, and make correct predictions of experiments. | |
| "The developed formalism2 adds to our understanding of physics of liquid crystal nanocolloids," he says. "This new understanding has important practical implications. Now we have a simple yet powerful tool to design, perform, and analyze experiments." | |
| "Moreover" he adds, "the proposed method can be useful in the design of highly efficient ion trapping nanomaterials." | |
| Having this understanding of the ion trapping phenomena in liquid crystals, the problem of their uncontrolled ionic contamination can be solved. | |
| Practical advice for scientists and R&D engineers: The ionic purity of nanoparticles should be controlled and the use of highly purified nanomaterials in liquid crystal research is a must in order to achieve a high level of purification. If this factor – the ionic purity – is not controlled by an experimentalist, the results can vary dramatically. | |
| The basic idea of Garbovskiy's paper in APL – the use of the Langmuir adsorption isotherm and the quantification of the nanoparticle's contamination by means of the dimensionless contamination factor – to describe possible effects of contaminated nanoparticles on the electrical properties of liquid crystals, laid the foundation for four subsequent publications3-6. | |
| A paper in Liquid Crystals3 is a logical extension of the APL paper and is aimed at covering in more detail the case of two types of ions (adsorbates) in liquid crystal nano-colloids. | |
| The second publication in Liquid Crystals4 is aimed to answer the obvious question of how well the approach proposed in the APL paper can describe actual experimental data. | |
| This question was fully addressed by applying the previously proposed formalism to a broad variety of existing experimental results. | |
| "This formalism successfully describes the dependence of the concentration of mobile ions in various liquid crystal nano-colloids on the nano-dopant?s loading: liquid crystals doped with anatase nanoparticles; liquid crystals doped with carbon nanotubes; liquid crystals doped with diamond nanoparticles; liquid crystals doped with graphene; liquid crystals doped with ferroelectric nanoparticles4," says Garbovskiy. | |
| He adds that the observed excellent agreement between simple approach and experimental results reported by independent research groups unambiguously points to the applicability of the proposed idea. | |
| The applicability of the Langmuir adsorption isotherm to describe the adsorption/desorption of ions in liquid crystals, size effects and possible impact of high electric field were explored further in another paper in Liquid Crystals5. A paper in Chemical Physics Letters6 addresses practical needs of R&D engineers by providing data on ion trapping coefficient of various nanomaterials in liquid crystals. | |
| Research on nanoparticles and ions in liquid crystals is very broad and involves many international research teams. | |
| "These research efforts cover a wide range of nanomaterials and different types of liquid crystals," notes Garbovskiy. "Intriguing aspects of this research are effects of external factors – electric and electromagnetic fields, temperature, acoustic impact etc. – on the ion trapping properties of nanoparticles dispersed in liquid crystals. A more general theory, yet to be developed, should also account for these possible effects." | |
| References | |
| 1) Y. Garbovskiy, I. Glushchenko, Nano-Objects and Ions in Liquid Crystals: Ion Trapping Effect and Related Phenomena, Crystals | |
| 2) Y. Garbovskiy, Switching between purification and contamination regimes governed by the ionic purity of nanoparticles dispersed in liquid crystals, Applied Physics Letters | |
| 3) Y. Garbovskiy, Impact of contaminated nanoparticles on the nonmonotonous change in the concentration of mobile ions in liquid crystals, Liquid Crystals | |
| 4) Y. Garbovskiy, Electrical properties of liquid crystal nanocolloids analysed from perspectives of the ionic purity of nano-dopants, Liquid Crystals | |
| 5) Y. Garbovskiy, Adsorption / desorption of ions in liquid crystal nano-colloids: the applicability of the Langmuir isotherm, impact of high electric fields, and effects of the nanoparticle's size, Liquid Crystals | |
| 6) Y. Garbovskiy, Adsorption of ions onto nanosolids dispersed in liquid crystals: towards understanding the ion trapping effect in nanocolloids, Chemical Physics Letters | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
