| Posted: Sep 27, 2016 | |
A nanobiotechnology tool for site-specific delivery in the gastrointestinal tract (w/video) |
|
| (Nanowerk Spotlight) Last year, researchers at the University of California, San Diego have shown that a micromotor fueled by stomach acid can take a bubble-powered ride inside a mouse. These tiny motors, each about one-fifth the width of a human hair, may someday offer a safer and more efficient way to deliver drugs or diagnose tumors (see our previous Nanowerk Spotlight: "First demonstration of micromotor operation and payload release in living organism (w/video)". | |
| Continuing this pioneering effort toward enhanced motor-based site-specific delivery, the research team, led by Professors Joseph Wang and Liangfang Zhang at the Nanoengineering Department at University of California, San Diego (UCSD), now have developed an enteric micromotor consisting of a magnesium (Mg)-based motor body with an enteric polymer coating. | |
| The new study has been published in the September 20, 2016 online edition of ACS Nano ("Enteric Micromotor Can Selectively Position and Spontaneously Propel in the Gastrointestinal Tract"). | |
 |
|
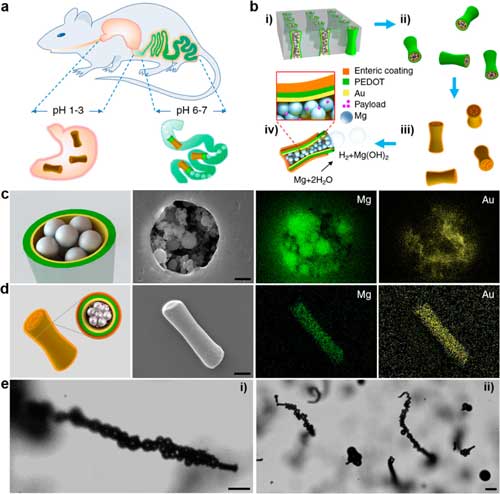
| Synthesis and characterization of enteric magnesium micromotors (EMgMs). (a) Schematic illustration of in vivo operation of the EMgMs for propulsion and localized delivery to the GI tract. (b) Preparation of EMgMs: (i) loading of Mg microspheres and payload into PEDOT/Au microtubes electrodeposited in microporous polycarbonate (PC) membrane with pore size of 5 µm and pore length of 15 µm; (ii) dissolution of PC membrane and release the Mg micromotors; (iii) coating Mg micromotors with enteric polymer; (iv) dissolution of the enteric coating and propulsion of Mg micromotors in solution with neutral pH. (c) Top view of a Mg micromotor with scanning electron microscopy (SEM) characterization and engergy-dispersive X-ray (EDX) images of the Mg and Au in the micromotor. Scale bar: 1 µm. (d) Side view of an EMgM with SEM characterization and EDX images of the Mg and Au in the micromotor. Scale bar: 5 µm. (e) Propulsion snapshot of a single (i) and multiple (ii) EMgMs in the intestinal fluid. Scale bars: 20 µm. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| "The new motors, aimed controlling and enhancing site-specific delivery in the gastrointestinal (GI) tract, consist of water-powered magnesium-based tubular micromotors coated with an enteric polymer layer," Wang explains to Nanowerk. "The microscale robot can deliver payload to particular location via dissolution of their enteric polymeric coating to activate their propulsion at the target site towards localized tissue penetration and retention." | |
| The enteric coating can shield the motors from an acidic gastric fluid environment (pH 1-3) but dissolves in intestinal fluid (pH 6-7) to expose the motors to their fuel and start the movement. | |
| Wang points out that, by simply tuning the thickness of the pH-sensitive coating it is feasible to selectively activate the propulsion, at desired regions of the GI tract, and thus to control their localized tissue penetration and retention. | |
| The team evaluated the properties and functions of the synthesized enteric magnesium micromotors in a mouse model. The in vivo results demonstrate that these motors can safely pass through the stomach and accurately activate in the GI tract without causing noticeable acute toxicity. | |
| "Our micromotor-based GI transporter system offers innovative combination of accurate positioning and active propulsion towards effective localized GI delivery and improved retention in the GI tract," notes Zhang. "These developments are of particular importance for the emerging microbiome research." | |
| This video illustrates the operation principle of enteric magnesium micromotors that can selectively position and spontaneously propel in the gastrointestinal tract by using the pH-sensitive coating that dissolves in intestinal fluid. (Video: UCSD) | |
| The use of advanced pH-sensitive materials for precise local manipulation of microrobot for site-specific active delivery – compared to conventional passive-diffusion-driven delivery vehicles – is expected to pioneer novel delivery approaches and advance the emerging field of medical nano/micromotors and nanorobotics. | |
| Future studies by the UCSD team aim at evaluating the delivery efficiency and enhanced therapeutic efficacy associated with the operation and propulsion of their nanomotor carriers. | |
| Similar nanobiotechnology nanomachines may find a variety of applications including diagnostics, drug delivery, or nanosurgery. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
