| Posted: Nov 15, 2016 | |
Impurities enhance the performance of perovskite solar cells |
|
| (Nanowerk Spotlight) The development of perovskite solar cells, first reported in 2009, is a possible route towards high efficiency photovoltaics that are also cost-effectiveness, owing to to their easy-processing from solution. They absorb light across almost all visible wavelengths, they have exceptional power conversion efficiencies exceeding 20% in the lab, and they are relatively easy to fabricate. Question marks have however remained on their stability. | |
| Researchers in Germany have now demonstrated in detail how chemical purity of perovskite precursors can affect the morphology of thin-film perovskite layers. | |
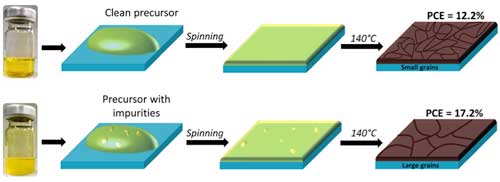
| While most studies concentrate on the exploration of processing conditions, the team has investigated the purity levels of common perovskite precursor solutions and found a number of impurities that are critically important toward controlling the crystallization of perovskites. | |
| They observed that impurities that formed during the preparation of perovskite precursor solutions initiate formation of PbHPO3 nanoparticles, which helps to growth larger perovskite crystal grains resulting in higher solar cells efficiency. | |
| They have reported their findings in Advanced Material Interfaces ("Deciphering the Role of Impurities in Methylammonium Iodide and Their Impact on the Performance of Perovskite Solar Cells"). | |
 |
|
| Schematic interpretation of the perovskite thin film growth based on different precursor solution with and without PbHPO3 nanoparticles. (Image: i-MEET) (click on image to enlarge) | |
| "We have also demonstrated that the increased perovskite grain size, the reduced defect density, and the enhanced intrinsic perovskite stability of the thin films are achieved by using perovskite precursor solution that contains PbHPO3 nanoparticles," Ievgen Levchuk, a PhD student in the group of Prof. Christoph J. Brabec (Materials for Electronics and Energy Technology – i-MEET) at Friedrich-Alexander-Universität Erlangen-Nürnberg, and first author of the paper together with co-author Yi Hou, a PhD student in the same group, tells Nanowerk. " Our findings indicate that the understanding and ability to control the amount of impurities in perovskite precursor solution is important for the full control and reproducibility of perovskite solar cell fabrication." | |
| The study highlights the chemical investigation of the possible impurities formed during methylammonium iodide (CH3NH3I) preparation and reveal their relevance during perovskite solar cell processing. | |
| The scientists found that the impurities are a side product during CH3NH3I synthesis in the presence of hypophosphorous acid (H2PO2) as a stabilizer for hydriodic acid, namely methylammonium hypophosphite (CH3NH3H2PO2) and methylammonium phosphite (CH3NH3H2PO3). | |
| "These contaminators initiate PbHPO3 nanoparticle formation in the perovskite precursor solution, with further seed growth during thin-film formation resulting in increased perovskite grain size, a reduced defect density, and an enhanced intrinsic perovskite stability," explains Levchuk. | |
| He points out that, as a result of the presence of impurities and by using a simple planar inverted (p-i-n) structure, the performance of perovskite devices could considerably be enhanced from 12.2% to 17.2% at roll-to-roll compatible processing temperature conditions (<140 °C). | |
| "Our findings indicate that the understanding and ability to control the amount of impurities in perovskite precursor solutions is extremely important for achieving full control and reproducibility of solar cell fabrication," Levchuk concludes. "Therefore, quality control for self-synthesized or commercially available CH3NH3I is necessary." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
