| Posted: Nov 17, 2016 | |
A new way to make nanostructured phosphides for efficient water splitting |
|
| (Nanowerk Spotlight) A major challenge in transitioning to a clean energy society is how to store excess electricity. Renewable energy sources such as solar, wind, hydroelectric and geothermal fluctuate over time, and storing this energy is a crucial task for creating a sustainable society. | |
| One of the most promising ideas is to use the excess electricity to generate chemical fuel via electrolysis, where the water is split into hydrogen and oxygen using an electrical current. The hydrogen can then be combined with oxygen from the atmosphere in clean fuel cells or with carbon dioxide in the oceans or atmosphere to create raw materials for hydrocarbon fuels and materials. | |
| Water electrolyzers usually use precious metals as catalysts (e.g. platinum or ruthenium oxide), limiting large-scale applications due to cost and scarcity. Most commercial hydrogen production therefore still uses fossil fuels such as natural gas, oil and coal, because this approach today is cheaper than electrolysis of water. | |
| Efficient water splitting for large-scale, industrial applications requires highly active, low cost, and robust catalysts. | |
| "Motivated by this challenge, enormous efforts have been devoted to developing cost-efficient alternatives including sulfides, selenides, phosphides, and many other nonprecious transition metal compounds," Husam N. Alshareef, a professor in material science and engineering at King Abdullah University of Science & Technology (KAUST) in Saudi Arabia, tells Nanowerk. "In our recent work, we have shown for the first time that metals or metal hydroxides can be rapidly converted into phosphides at low temperature using a newly developed plasma-assisted route." | |
| Commonly practiced methods suffer from high temperature and long reaction time and there is a need to develop facile and effective methods to synthesize phosphides. | |
| Using combined theoretical and experimental studies, Alshareef's team established that NiCoP, a rarely explored ternary compound, can achieve stable catalytic performance toward both the hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) that is superior to that reported for metal phosphides and metal chalcogenides, and is among the most active water splitting electrocatalysts reported so far. | |
| The researchers published their findings in the November 9, 2016 online edition of Nano Letters ("Plasma-Assisted Synthesis of NiCoP for Efficient Overall Water Splitting"). | |
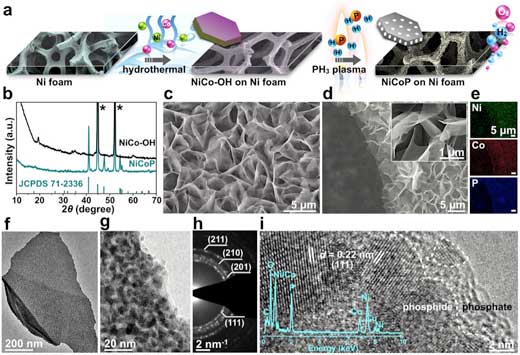 |
|
| Synthetic route and structural characterization of the NiCoP nanostructure. (a) Schematic illustration of the synthetic route for NiCoP nanostructure on Ni foam. (b) PXRD patterns of NiCo-OH and the converted NiCoP. The asterisks mark the diffraction peaks from Ni foam. (c) SEM image of NiCo-OH. (d) SEM images and (e) corresponding EDS elemental maps of the NiCoP. (f,g) TEM images, (h) SAED pattern, and (i) high-resolution TEM image and EDS spectrum (inset) of the NiCoP. The dashed white line highlights the crystalline-amorphous boundary. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| "Our as-prepared NiCoP nanoparticles with rich defects exhibit extremely low overpotential for the overall water splitting electrocatalysis in alkaline solutions along with outstanding stability and is among one of the most active electrocatalysts," notes Dr. Hanfeng Liang, post-doctoral researcher in Alshareef's group and lead author of the study. | |
| The team's newly developed plasma-assisted method can serve as a versatile route to synthesize various nanostructured metal phosphides. Furthermore, their new concepts on tuning the electronic structure and reactivity by substituting metal atoms in binary metal phosphides could be generally applicable for enhancing electrocatalytic applications. | |
| Commonly practiced processes for the preparation of metal phosphides can be generally divided into two categories: the vapor phase method and the solution phase method. | |
| The former requires high reaction temperature (normally between 300-700°C, depending on the decomposition temperature of the phosphorus sources) and long reaction time (several hours). Whereas the latter often involves the use of organic phosphorus sources, typically tri-n-octylphosphine (TOP). | |
| "The drawback is that the insolubility of TOP (and other commonly used organic phosphorus sources) in water and its high decomposition temperature restricts the system to be only conducted in high boiling-point organic solvents, leading the reaction to be highly flammable and corrosive and should be carried out under severely oxygen free conditions by skilled workers," explains Liang. "Our newly developed method utilizes the high reactivity of plasma and is able to synthesize monometallic, bimetallic, or even more complex phosphides at lower temperatures (100-250°C) in a rapid manner (30 seconds to 20 minutes), which significantly improves the efficiency and lowers the cost." | |
| He continues: "The Gibbs free energy for the adsorption of reaction intermedia can be used to evaluate the activity of catalysts. Taking HER as an example, since it relies on the reversible adsorption and desorption of hydrogen (H), ideally the H binding should be neither too strong nor too weak, that is, the Gibbs free energy of the adsorbed H* (* being the adsorption site) should be close to thermo-neutral (|ΔGH*| = 0). | |
| "From this viewpoint, the optimal catalytic activity could be achieved by optimizing the surface adsorption energy. Using NiCoP as a demonstration, we showed that the Co substitution could effectively tune the electronic structure and significantly lower and the surface H adsorption energy of Ni2P, and thus greatly boost the catalytic activity. The resulting NiCoP exhibited extremely low onset overpotentials toward both the hydrogen evolution reaction (-32 mV at -10 mA cm-2) and oxygen evolution reaction (280 mV at 10 mA cm-2) in alkaline media." | |
| This activity places NiCoP among the best Earth-abundant water splitting electrocatalysts reported to date. | |
| The team's next goal is to gain a better understanding of the detailed catalytic mechanism and to further integrate this compound with semiconducting materials for photoelectrochemical water splitting devices. They are also planning to synthesize new phosphide for electrochemical energy storage devices including batteries and supercapacitors. | |
| Furthermore, even though NiCoP showed decent catalytic performance toward the OER, the origin of the high activity is not fully understood. A theoretical calculation could be helpful for revealing the science behind it. | |
| "To date, the developed HER catalysts normally work in acidic media, whereas the OER is generally conducted in alkaline solutions since most electrocatalysts cannot survive in acid under strong oxidative conditions of OER," concludes Alshareef. "Such pH mismatch becomes a big problem when assembling a water splitting device with both HER and OER catalysts and often leads to poor overall performance. In view of this, an important future direction would be to develop stable bifunctional electrocatalysts that can efficiently catalyze both the HER and OER in the same electrolyte." | |
| "Another particular challenge is that currently there is still a lack of theoretical prediction that can better guide the efficiently screening, selection, and further rational design of new materials or compounds for electrocatalysis." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
