| Posted: Feb 09, 2017 | |
Two-dimensional oxides juice up sodium ion batteries |
|
| (Nanowerk Spotlight) Sodium ion batteries are a promising alternative to lithium ion batteries, particularly for home based and grid level storage solutions. Despite its lower energy density, sodium is more abundant and geographical more evenly distributed than lithium – and it is cheaper. In some countries, such as Saudi Arabia, it is abundant as a natural product of the water desalination process. | |
| Tin monoxide (SnO) has been demonstrated to have excellent physical and chemical properties and has a large theoretical capacity as battery anode, for instance for sodium ion batteries. | |
| Unfortunately, though, it also exhibits large volume change during the sodiation and lithiation process (> 300%), which makes it unsuitable as a high-performing anode material. | |
| Researchers have attempted various ways to mitigate volume change in this important material. For instance, SnO material has often been mixed with different forms of carbon to mitigate its volume change. | |
| A new strategy from the labs of Prof. Husam Alshareef at King Abdullah University of Science & Technology (KAUST) is different. | |
| "Recognizing that SnO actually has a two-dimensional structure, we set out to find a chemical process to control the degree of exfoliation of this oxide during chemical synthesis," Alshareef tells Nanowerk. "By carefully choosing the reactants, solvents, and reaction conditions, we found a way to precisely control the number of atomic layers in each SnO sheet. In fact, we could easily go from just a couple of atomic layers to 20 atomic layers in each SnO sheet." | |
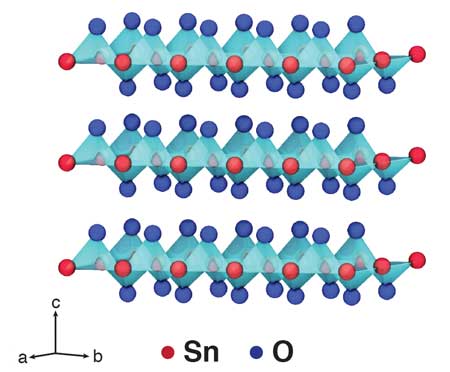 |
|
| Schematic illustration of the crystal structure of two-dimensional SnO. (Image: KAUST) | |
| This study, which the team has reported in Nano Letters ("Two-Dimensional SnO Anodes with a Tunable Number of Atomic Layers for Sodium Ion Batteries"), represents a different approach for designing the SnO anodes and mitigating their volume change. | |
| Although SnO has a layered structure, studies on exfoliating it into individual atomic layers to control volume change during battery operation have not been reported so far. | |
| The KAUST team demonstrated that, when the number of SnO atomic layers in one SnO nanosheet is less than five, the sodium ion batteries could last for thousands of cycles. | |
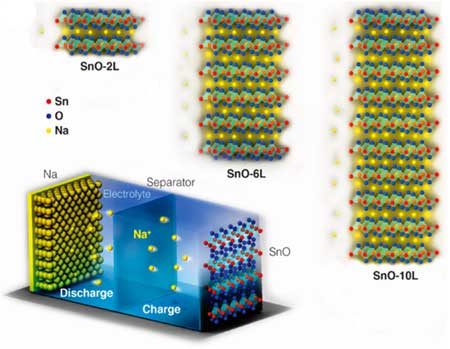 |
|
| Schematic illustration of the SnO nanosheets with different number of layers and the half-cell structure. (Reprinted with permission by American Chemical Society) | |
| "The sodiation density was 452 mAh/g after 1000 cycles and showed no sign of instability, which is one of the best reported performance for this anode material," says Fan Zhang, PhD student and lead author of the study. "With our process, the volume change is largely mitigated due to the atomic scale thickness of the SnO sheets." | |
| "We have been focussing on tin-based anodes in our group," adds Alshareef. "The results in this present work are one of the best results we have obtained for this oxide in sodium ion batteries." | |
| "The natural next step is to make full-cell batteries with the appropriate cathode materials," he concludes. "The combined effects of cathode material type, electrolyte, and separator used should be evaluated since they also may affect the overall performance of the battery. The challenge is to optimize the other components of the battery to leverage the full potential of the SnO anodes. Once the full cells are optimized, larger batteries and even battery packs could be constructed and tested." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
