| Posted: Mar 28, 2017 | |
Improving Li-ion batteries with nanostructured conductive polymer gels |
|
| (Nanowerk Spotlight) The electrode in lithium-ion (Li-ion) batteries is an integrated system in which both active materials and binder systems play critical roles in determining its final properties. In order to improve battery performance, a lot of research is focussing on the development of high-capacity active materials. However, without an efficient binder system, these novel materials can't fulfill their potentials. | |
| A group of researchers has contributed to this field from a slightly different aspect. They developed an entirely new binder system with a nano-architecture that promotes both electron and ion transport, which enhances the energy per unit mass and volume of the electrode. | |
| This work by Guihua Yu's group at University of Texas at Austin and Esther Takeuchi's group at Stony Brook University, demonstrates a new generation of nanostructured conductive polymer gel based binder materials for fabrication of high-energy lithium-ion battery electrodes. This gel framework could become a next-generation binder system for commercial Li-ion batteries. | |
| "Compared to a conventional binder system, which typically consists of conductive additive and polymer binder, our novel binder plays dual functionalities simultaneously by combining conductive and adhesive features,"Professor Yu tells Nanowerk. "More importantly, owing to its unique 3D network structure, this gel binder promotes both electron and ion transport in electrodes and improves the distribution of active particles, thus enhancing their rate performance and cycle life." | |
| He points out that this invention is important because it presents a new generation of powerful yet scalable binder materials for lithium ion batteries that show great potential in industrial manufacturing. | |
| The researchers have reported their findings in two papers in Nano Letters ("Nanostructured Conductive Polymer Gels as a General Framework Material To Improve Electrochemical Performance of Cathode Materials in Li-Ion Batteries") and Advanced Materials ("A Tunable 3D Nanostructured Conductive Gel Framework Electrode for High-Performance Lithium Ion Batteries"). | |
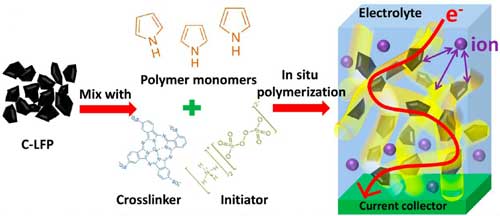 |
|
| Schematic of synthetic and structural features of commercial lithium iron phosphate (C-LFP)/cross-linked polypyrrole (C-PPy) hybrid gel framework. The conductive polymer chains can be polymerized in situ with electrode materials and cross-linked by molecules with multiple functional groups, resulting in a polymeric network connecting all active particles. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| "A traditional binder system in Li-ion battery electrodes is a binary hybrid with its components performing separate functions," explains Yu. "In such a system, polymer binders like polyvinylidene fluoride (PVDF) keep the active materials and other additives together to assure mechanical integrity, while a conductive additive (usually carbon particles) ensures the conductivity of the entire electrode." | |
| In these electrodes, electrons transport through chains of particles while ions move through the liquid or solid electrolyte that fills the pores of the electrode. However, the conductive phases are randomly distributed, which may lead to bottlenecks and poor contacts that impede effective access to parts of the battery. | |
| And both organic and inorganic components tend to aggregate, which also negatively impact electron and ion transport. | |
| The team's novel conductive gel binder can overcome these drawbacks to improve the rate and cyclic performance of Li-ion batteries. | |
| The conductive polymer gels potentially could also be used for responsive/smart electronics such as biosensors, artificial skins and soft robotics. | |
| "Firstly, our work provides a new method for synthesis of conductive polymer gels," elaborates Yu. "Traditionally, conductive polymer gels are synthesized by template-based method, which usually results in low conductivity and poor mechanical properties. The method we developed is to crosslink conductive polymer chains with functional molecules with multiple functional groups, enabling a network structure promoting high electronic conductivity and electrochemical activity." | |
| "Secondly, we demonstrated that this newly developed conductive polymer gel can be used as binder system to significantly improve conventional lithium-ion battery performance," he continues. "The ease of processability and excellent chemical and physical properties of these nanostructured conductive gels enables a new class of binder materials for fabricating next-generation, high-energy lithium-ion batteries." | |
| Although the researchers' binder gel is mechanically strong, it lacks flexibility and stretchability. The plan is to further modify the mechanical properties by tailoring the molecular backbones of conductive polymers through the addition of side chains or other building block polymers. | |
| The scientists further intend to demonstrate the versatility of their gel binders for other important electrode materials, such as some commercial electrode materials as well as some next-generation ultrahigh-capacity materials, such as silicon, and sulfur. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
