| Posted: Jun 22, 2017 | |
Enhancing radiation therapy with nanotechnology |
|
| (Nanowerk Spotlight) In an effort to devise novel and more effective anticancer regimes, a rapidly growing community of researchers is applying the unique properties of nanomaterials to combat the unmet challenges posed by classical radiation therapy (RT) – which has become one of the most effective and frequently applied cancer therapies. | |
| The high surface area, stability, and facile tunability of nanomaterials make them ideal for transportation of chemotherapeutics, phototherapeutic agents, radiosensitizers, oxygen reservoirs, etc. across several physiological barriers. | |
| However, despite a plethora of preclinical studies, nanoparticle mediated combination RT/phototherapy has not yet been translated in the clinic. | |
| A major advantage of RT lies in its noninvasive nature, which leads to less physiological and psychological burden being placed on the patients. However, collateral damage to healthy tissue resulting from its nonspecific nature means only limited doses can be administered, which, in turn, may result in tumor recurrence from a surviving population of radioresistant cells. | |
| In a recent Perspectives article in ACS Nano ("Harnessing the Power of Nanotechnology for Enhanced Radiation Therapy"), researchers from the University of Wisconsin take a brief look at the emerging roles of nanotechnology in the rapidly evolving domain of modern/future radiation therapy. | |
 |
|
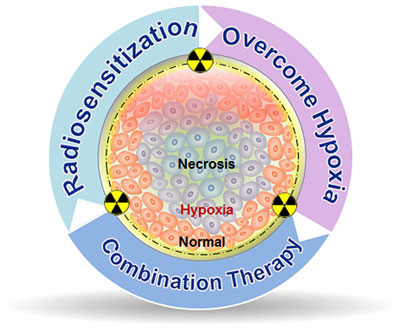
| The article reviews the emerging technologies for optimization of radio sensitization, hypoxia modulation, and combinatorial therapeutic regimes for improved treatment outcomes in preclinical tumor models, with a focus on nanotechnology-mediated approaches. (© ACS) | |
| The authors stress that several key points need attention as the field progresses: | |
| First, there is a pressing need for rationally designed nanoformulations that exhibit optimal tumor-homing and intratumor distribution. Improved tumor penetration by such nanoformulations can enhance RT outcomes, especially when combined with hypoxia-responsive therapies. The complex tumor microenvironment undergoes dynamic interactions with administered nanoparticles and RT, which can drastically influence treatment outcomes and, thus, warrant careful consideration during therapy planning. | |
| Second, a paucity of data on the long-term in vivo behavior and toxicity profiles of nanoparticles has severely impeded the transition of even well-established nanoformulations from preclinical to clinical settings. Despite sophisticated designs and excellent preclinical performance, most nanoparticles employed for RT to date demonstrate suboptimal pharmacokinetics in vivo, leading to grave toxicity concerns and even rejection of such 'fantastical studies' by many in the medical community. Although innovative technologies are important for moving the field forward, in-depth evaluation of their interactions with tissues and cells is certainly warranted. | |
| Third, nanotechnology provides a promising platform to bridge the gap between the newly emerging targeted therapies and traditional RT. For example, immunotherapy has generated much excitement in the medical community owing to its exemplary results in melanoma patients. | |
| Nanoparticle-based radiosensitizers designed to deliver alternative payloads such as immune adjuvants as well as angiostatic drugs and antibodies are interesting avenues to explore. | |
| Finally, concerted efforts on the part of scientists and regulatory authorities are required to expedite the translation of promising, clinically beneficial nanomaterials from bench to bedside. | |
| "Continued support and investment from governments and other funding agencies is needed for cancer nanotechnology-related research before we can harness the full power of this exciting technology and reap its fruit for future precision cancer therapy in the clinic to benefit millions of patients," the authors conclude. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
