| Posted: Jul 04, 2017 | |
Integrating biomolecules with metal-organic frameworks |
|
| (Nanowerk Spotlight) Metal-organic frameworks (MOFs) are well-ordered, lattice-like crystals. The nodes of the lattices are metals – such as copper, zinc, nickel or cobalt – which are connected by organic molecules. | |
| The most impressive features of MOFs are their extremely high surface area (up to 10000 m2 g-1), high porosity (up to 90%) and tunable pore sizes, which are remarkable advantages over other porous materials (e.g., zeolites and carbons). | |
| With their special structure and large surface area, MOFs open up new opportunities for alternative systems for gas and energy storage (e.g. carbon dioxide and hydrogen storage), separation, in catalysis, chemical sensing, as nanoreactors, and in drug delivery. | |
| The ability to exchange the metal centers and organic linkers even provide an extensive library of MOF materials. As a result, the integration of small guest molecules within the MOF pores, such as small molecule drugs and biomolecules, have shown promise for delivery applications to treat diseases. | |
| Several of the key inherent properties of MOFs make them great candidates for such drug delivery systems: Firstly, there are multiple pathways to incorporate guest molecules within the MOFs and secondly, the tailorable interactions between the guest molecules and MOF linkers or metal nodes allow for high drug loading capacity. | |
| Additionally, the tunable sizes of MOF crystals enhance optimization for cellular uptake, the potential for chemical-environment-dependent dissociation of MOF species for stimuli-responsive and triggered guest release; as well as the biocompatibility of certain classes of MOFs at uptake concentrations. | |
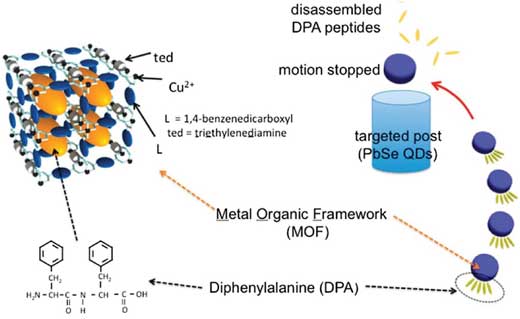 |
|
| pH gradient change in environment triggers the disassembly of diphenylalanine from the MOF and particle movement. (©American Chemical Society) | |
| A recent review article in Small ("Integration of Biomolecules with Metal–Organic Frameworks") discusses current proceedings on integrating diverse biomolecules within MOFs. | |
| The authors, from the Tsung Research Group at Boston College, first discuss works on incorporating relatively small and simple biomolecules including lipids, oligopeptides, and oligonucleotides, followed by works performed for integrating more complex proteins. | |
| They then undertake a focus on the methodology development of biomolecule incorporation and share the prospective views of material synthesis. | |
| Discussing some of the current challenges on the integration of biomolecules into MOF structures – loading optimization, biomolecule preservation, MOF biocompatibility – they write about the limitations and omissions of the reported works, as well as possible ways to overcome these problems. | |
| Biomolecule-integrated MOF materials have many potential applications. Molecule delivery might be the most possible application for biomolecule-MOF complexes, where MOF particles can serve as carriers for biomolecules as payloads. | |
| Biomolecule-integrated MOF particles make the delivery of some vulnerable biomolecules such as nucleic acids and proteins possible because they can shield and protect the integrated biomolecules from ambient disturbance, while also providing enough interior space for the high-efficiency biomolecule loading. | |
| After successful delivery, biomolecules can be unloaded to fulfill tasks such as photo dynamic therapy, protein regulation, and RNA interference (RNAi). | |
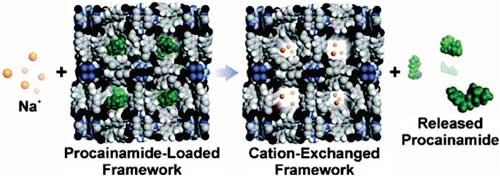 |
|
| Cation-triggered drug release from bio-MOF-1. (©American Chemical Society) | |
| Molecule detection is another possible application. Integrated biomolecules can exchange and interact with exotic species such as small molecules and ions which helps read out the target signals. Here, MOF particles are used as mini platforms for target capture and enrichment. Other possible applications include targeting enhancement gained from conjugated outside biomolecules, intracellular imaging based on intracellular biomolecule confinement. | |
| "However, despite the recent exciting proceedings in biomolecule-integrated MOF hybrid materials, there still lie some problems in the field," the authors conclude their review. "The loading optimization, the biomolecule protection, and the MOF biocompatibility were all briefly discussed, but more questions needed to be considered. Nevertheless, this high potential field is a relatively new field with several exciting areas to explore." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
