| Posted: May 05, 2006 | |
Combining gold nanoparticles with viruses for a combined thermal/gene cancer therapy |
|
| (Nanowerk News) Gold nanoparticles have shown promise for cancer therapy by virtue of their ability to absorb laser light, heat up and thus kill the tumor. However, in order for the gold nanoparticles to have their therapeutic effect they need to be taken up specifically by tumor cells. Researchers have now used targeted viral vectors as carriers to bring the gold nanoparticles to the location where they need to work. This opens up the possibility of combining two types of treatment: heating the tumor using the gold nanoparticles, and killing the tumor using gene therapy. | |
| An international group of researchers led by scientists from the Gene Therapy Center at the University of Alabama (UAB) recently published findings that indicate the possibility of using adenoviral vectors as carriers for gold nanoparticles. Their paper, titled "Covalently Linked Au Nanoparticles to a Viral Vector: Potential for Combined Photothermal and Gene Cancer Therapy" was published on March 15, 2006 in the online edition of Nano Letters. | |
| Dr. Maaike Everts from UAB, first author of the paper, explained their findings to Nanowerk: "We found that coupling gold nanoparticles to the surface of viruses that are targeted to tumor cells allows them to ‘hitchhike’ along with the virus to the tumor, without impeding the ability of the virus to infect and target tumor cells." | |
| Dr. David T. Curiel, who heads the Gene Therapy Center, and his team are developing cancer gene therapy approaches by using viruses, in particular adenovirus. Adenovirus causes the common cold, but the particular viruses Curiel and his group use have been genetically altered in order to stop them from multiplying, thus rendering them harmless to the human body. | |
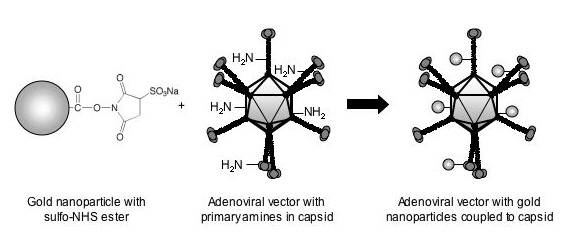 | |
| Gold nanoparticles can covalently be attached to primary amines present in capsid proteins. The sulfo-NHS ester attached on the 1.3-nm gold particle will react with primary amines of lysine residues present in the adenoviral capsid, resulting in an amide bond between the gold particle and the adenovirus. There are over 10 000 lysines in the proteins that make up the capsid, although not all of these will be accessible for chemical reaction.. (Reprinted with permission from ACS) | |
| "We are able to target these viruses – also known as ‘viral vectors’ in a gene therapy context – to tumors by virtue of specific markers that are expressed on cancer cells, which can be recognized by our viruses" Curiel says. | |
| Previously, the payload of a viral vector was either a corrective gene (an example is a correct copy of the p53 gene, which is mutated in a lot of cancers) or a gene that encodes a toxic product that is able to kill the tumor cell. | |
| "The novelty in our work," says Everts "is the use of metal nanoparticles as a payload of targeted viral vectors. Since the viruses are still able to deliver a gene of interest, even with the gold nanoparticles coupled to their surface, a combination of both payloads is a possibility, which may work better than either alone." | |
| In addition, although gold nanoparticles have been targeted to tumor cells before, using antibodies or peptides for example, the method of targeting them to tumors by using viral vectors is a novel approach. | |
| One of the declared goals of the NCI Alliance for Nanotechnology in Cancer is the development of multifunctional nanoparticles: an “all-in-one toolkit” that can 1) target tumor cells specifically; 2) image the tumor using non-invasive methods such as MRI or PET; 3) treat the tumor and 4) give feedback whether the treatment has worked or not. The UAB scientists now have have demonstrated that they can combine 1) and 3) thus far. | |
| "We are working on strategies to incorporate modalities 2) and 4) as well, but there is still a lot of work that needs to be done before that can be accomplished" Everts says. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
