| Posted: Mar 06, 2008 | |
Catching a rainbow - quantum dot nanotechnology brightens the prospects for solar energy |
|
| (Nanowerk Spotlight) Harnessing the power of the sun to replace the use of fossil fuels holds tremendous promise. One way to do this is through the use of solar, or photovoltaic, cells. Large-scale installation already show the technical feasibility of this technology although the major problem of photovoltaic solar energy - its relative inefficiency - still needs to be overcome to make the cost of electricity produced by solar cells equal or less than electricity produced by nuclear or fossil fuels. Until now, solar cells that convert sunlight to electric power have been dominated by solid state junction devices, often made of silicon wafers. Efforts are being made in laboratories worldwide to design ordered assemblies of semiconductor nanostructures, metal nanoparticles and carbon nanotubes for constructing next generation solar energy conversion devices. | |
| Quantum dots have been identified as important light harvesting material for building highly efficient solar cells. Quantum dots are nanoscale semiconductor structures which, when exposed to light at certain wavelengths, can generate free electrons and create an electrical current. Quantum dot technology represents an exciting field of research in solar energy yet the actual research results to use them in solar cells are relatively limited. | |
| By combining spectroscopic and photoelectrochemical techniques, researchers now have demonstrated size-dependent charge injection from different-sized cadmium selenide (CdSe) quantum dots into titanium dioxide nanoparticles and nanotubes, showing a way to maximize the light absorption of quantum dot-based solar cells. Termed 'rainbow solar cells', these next-generation solar cells consist of different size quantum dots assembled in an orderly fashion. Just as a rainbow displays multiple colors of the visible light spectrum, the 'rainbow solar cell' has the potential to simultaneously absorb multiple wavelengths of light and convert it to electricity in a very efficient manner. | |
| "One of the most important things we show in our new study is that one can use the same material (here, cadmium selenide quantum dots) to collect light across much of the solar spectrum" Dr. Prashant V. Kamat tells Nanowerk. "Because the sun emits light at a variety of wavelengths, it is important to be able to collect as many of those wavelengths as possible to maximize solar cell efficiency. Cadmium selenide quantum dots have long been known to collect light at multiple wavelengths, and have also previously been utilized in solar cells. However, this is the first study which directly compares how different sized quantum dots which absorb different wavelengths perform when incorporated into solar cells." | |
| Kamat, a professor of Chemistry & Biochemistry at Notre Dame University in Indiana, and a senior scientists at the university's Radiation Laboratory, and his team found that the smallest quantum dots (absorbing the shortest wavelengths of the solar spectrum) perform the best because they move electrons through the cell (i.e. create current) at the fastest rate. | |
 |
|
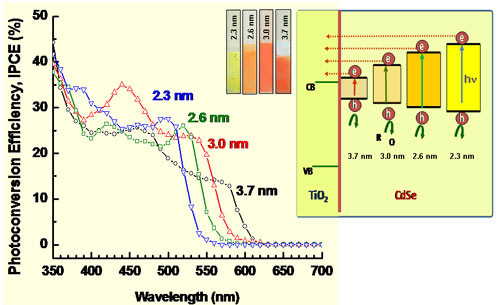
| Left: Photocurrent action spectra recorded in terms of incident photon to charge carrier generation efficiency (IPCE) of optically transparent electrodes/nanoparticulate titanium dioxide/CdSe electrodes. The individual IPCE responses correspond to (a) 3.7, (b) 3.0, (c) 2.6, and (d) 2.3 nm diameter CdSe quantum dots anchored on nanostructured TiO2 films. Right: Schematic diagram illustrating the energy levels of different-sized CdSe quantum dots and TiO2 (Image: Dr. Kamat, University of Notre Dame) | |
| This work, reported in the March 1, 2008 online edition of the Journal of the American Chemical Society ("Quantum Dot Solar Cells. Tuning Photoresponse through Size and Shape Control of CdSe-TiO2 Architecture"), both advances the general understanding of the nature of quantum dots, as well as provides information on how one can construct a better solar cell. Kamat's findings are threefold: | |
|
|
|
| Kamat tells us that his group was motivated to conduct this work for two specific reasons. "First, as a research group we recognize the scientific promise that quantum dots offer, and are interested in studying their properties and adding to a rapidly growing body of research which attempts to both better understand their physical behavior and find new uses in an attempt to improve the daily life of an average person. | |
| "Secondly, we are interested in laying the foundations for the best possible solar cell. As a research group, we are aware of the growing energy crisis and the need for the advancement of alternate forms of energy in the near future. The entire research group at Notre Dame is proud to be working on a problem – the improvement of solar cells – which society needs solved now more than ever." | |
| According to Kamat, the direct application of this work is to create the most efficient solar cell possible. In order to do this, the researchers need to accomplish three things: | |
| 1) collect as much of the solar spectrum as possible (accomplished here by using multiple sizes of quantum dots); | |
| 2) make sure that once such light is absorbed that it is converted into moving electrons (charge separation); and | |
| 3) once the electrons are created, make sure that they have the ability to freely move (charge transport) throughout the cell which creates electrical current. | |
| "While all three of these steps must be present to create the best possible solar cell, often times researchers study each step individually to isolate and improve upon any potential problems, with the hopes of eventually incorporating such improvements into a future solar cell" says Kamat. "One way in which our recent findings improve upon the third condition was by utilizing a tubular titanium dioxide base instead of a spherical titanium dioxide base. This advanced architecture allows electrons generated by the quantum dots to travel more freely within the cell because a tube provides an elongated direct pathway for electron travel whereas multiple spheres permit electron travel only if the electron "hops" between them, potentially slowing electron transport and hindering overall cell efficiency." | |
| Kamat says that, although the construction of this type of next generation solar cell is still in the works – his team is currently fine-tuning the engineering behind the construction – the theoretical framework behind this technology is sound: "Through the use of quantum dots as solar light absorbers, fabrication of 'rainbow solar cells' is on the horizon." | |
| We previously reported on Kamat's work on nanostructured solar cells in a Spotlight last year – Carbon nanotubes can double the efficiency of photoelectrochemical solar cells. | |
| Prof. Kamat's research is supported by Basic Energy Sciences of the US Department of Energy. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
