| Posted: Dec 20, 2017 | |
Voltage-activated carbon monoxide sensor |
|
| (Nanowerk Spotlight) The detection of carbon monoxide (CO) in the air is a vital issue, as CO is a highly toxic gas and an environmental pollutant. CO typically derives from the incomplete combustion of carbon-based fuels, such as cooking gas and gasoline; it has no odor, taste, or color and hence it is difficult to detect. | |
| Carbon monoxide is poisonous because it blocks the binding site for oxygen in hemoglobin. It is exactly this principle – a porphyrin ring with a central iron or cobalt atom that the poisonous gas attaches to – that can be used to implement sensors to warn against carbon monoxide. | |
| While carbon monoxide sensors have been extensively researched and commercialized, none make use of a molecule that can be voltage-activated using a newly reported method in Angewandte Chemie International Edition ("Bio-Inspired Carbon Monoxide Sensors with Voltage-Activated Sensitivity"). | |
| "We mimic the strong specific interactions between red blood cells and carbon monoxide to create a highly selective and customizable sensor," says Vera Schroeder, a Ph.D. candidate in the Swager Group at Massachusetts Institute of Technology (MIT), who, together with Suchol Savagatrup, is first author of the paper. "The molecules that detect carbon monoxide in our sensors can be activated to enhance their sensitivity. This activation changes the redox (reduction-oxidation) states of the molecules and is likely to have general applicability in chemical sensors and other fields." | |
 |
|
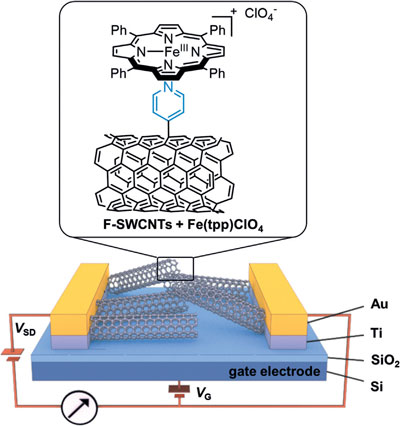
| Carbon monoxide detection by a bio-inspired sensor. Schematic of a field-effect transistor (FET) substrate with Au source-drain electrodes and Ti adhesion layer deposited on SiO2 dielectric layer and Si gate electrode. Chemical structures of pyridyl-functionalized single-walled carbon nanotubes (F-SWCNTs) and iron porphyrin (Fe-(tpp)ClO4), depicting the coordination chemistry of the pyridyl group to the iron center of the porphyrin. (Reprinted with permission by Wiley-VCH Verlag) | |
| Of course, a variety of CO detectors are commercially available, but there remains a need for massively distributed sensors that are small and inexpensive to prevent poisoning in domestic and industrial environments. | |
| To fabricate their sensor, the research team from MIT and King Abdullah University of Science and Technology (KAUST) applied a recently developed scheme for iodonium functionalization ("Covalent Functionalization of Carbon Nanomaterials with Iodonium Salts") to precisely attach single aromatic rings to the sidewalls of single-walled carbon nanotubes (SWCNTs). | |
| "This method allows us to confidently install a pyridyl group attached in the 4 position to the SWCNT for anchoring of the 5,10,15,20-tetraphenyl-porphyrin iron(III) perchlorate that serves as our redox active CO binding site," Schroeder explains to Nanowerk. | |
| Modulation of the gate voltage offers a predicted extra dimension for sensing. Specifically, the sensors show a significant increase in sensitivity toward CO when negative gate voltage is applied. | |
| The ability to predictively modulate the selectivity and sensitivity provides much more information from just one single sensor. | |
| Creating a practical sensor requires understanding of the specific chemical interactions. The team are seeking other examples of smart molecules that can be activated in a similar way for the detection of other harmful gases. In addition, they are working closely with electrical engineers to incorporate their sensors into portable devices. | |
| In the not too distant future we are likely to see chemical sensors of this type being integrated into our smartphones and other consumer electronics. | |
| "We are working towards sensors that can generate real-time and spatially-resolved information about our chemical environment," Schroeder concludes. "In a way, we are trying to give consumer electronics an additional sense; microphones allow hearing, cameras generate sight, capacitive sensors mimic physical touch, and chemical sensors will be able to impart taste and smell." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
