| Posted: Mar 26, 2018 | |
DNA origami as a novel avenue for creating next-generation optical voltage sensing nanodevices |
|
| (Nanowerk Spotlight) The term membrane potential refers to the electrical potential difference between the inside of a cell and its surrounding extracellular fluid. This phenomenon is present in all cells but it is especially important in nerve and muscle cells; in these cells, the changes in membrane potentials are used to code and transmit information. | |
| There are numerous biomedical topics that could be understood better by having more effective sensors for accurately quantifying the potential across cellular membranes. For instance, Alzheimer's disease has been linked to disrupted end-plate potential activity (i.e. the chemically induced change in electric potential of the motor end plate, the portion of the muscle-cell membrane that lies opposite the terminus of a nerve fibre at the neuromuscular junction). | |
| Apart from neuro-degenerative diseases, the areas that could benefit include muscular processes, the effect of neurotoxins and poisons on organisms, and simulating brain activity for the purposes of developing artificial intelligence. | |
| Over the past few decades, researchers have developed various optical voltage sensing probes in order to overcome the highly invasive nature of electrode-based techniques. | |
| "These voltage sensing mechanisms can be hampered by some combination of limitations including low sensitivity, slow kinetics, or heavy capacitive loading," Ulrich F. Keyser, a Professor of Applied Physics in the Cavendish Laboratory at the University of Cambridge, tells Nanowerk. "This has motivated our recent efforts to explore DNA nanotechnology for developing novel optical voltage sensing nanodevices that convert a local change of electric potential into optical signals." | |
| In a paper in Nano Letters ("Optical Voltage Sensing Using DNA Origami"), a research team, led by Keyser, Philip Tinnefeld from the Institute of Physical and Theoretical Chemistry at Technical University Braunschweig, and Aleksei Aksimentiev from the University of Illinois at Urbana-Champaign, has now reported for the first time, that a voltage can be read out in a nanopore with a dedicated Förster resonance energy transfer (FRET) sensor on a DNA origami. | |
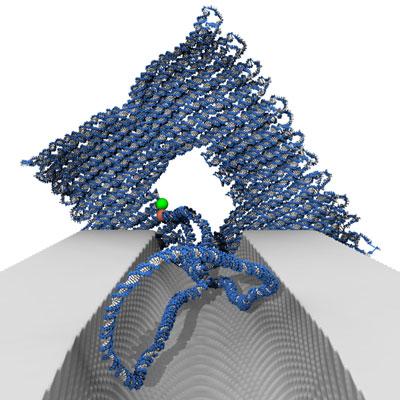 |
|
| View of the DNA origami voltage sensor through the nanochannel. (Image: C. Maffeo, UIUC) | |
| "We provide a method for sensing the difference in potential across a membrane by use of a simple single nanoscale sensor and light spectroscopy (fluorescence), which is a non-contact and sensitive technique," explains Keyser. "We wanted to show that voltage changes can be measured with a single FRET pair. The fact that we have single-sensor sensitivity, alongside the small size of structures, means that high spatial and high voltage resolution are possible, making possible the sensing of threshold voltages in real-time." | |
| The team used DNA origami as the building material for the sensor, which is a simple technique and allows the structure and size of the sensor to be adapted for different environments and situations. | |
| DNA works well as a material for voltage sensing as it is inherently charged and therefore can be manipulated to respond to changes in the surrounding potential. Specifically, the voltage-responsive, fluorescent DNA origami structure demonstrated in this work undergoes controlled structural deformation upon applying an electric field. This changes the distance between two light-sensitive molecules used in the FRET sensor and the correspondent signal can be read out optically. | |
| Simulation of the DNA origami voltage sensors and fluctuating dye molecules at 150 mV. (Animation: C. Maffeo, UIUC) | |
| "This is exciting as, with our work as a basis and proof-of-concept, there are many possibilities of adapting the design to increase its sensitivity and therefore furthering its potential applications," Keyser points out. | |
| The flexibility of DNA nanoassembly will allow the researchers to target their sensor design on a case-by-case basis. In addition to the programmability of the overall shape of the DNA-templated structure, the key strength of this technique lies in the ability to attach various chemical functionalities – fluorescent dyes, cholesterol moieties, or proteins – with base-pair precision. | |
| This feature allows for easy and quick adaptation of designs to a particular experimental requirement. Eventually this could lead to mass production of different types of sensors. | |
| An important aspect of this novel system is that, compared to ontogenetic approaches, it could be added externally to cells – i.e. there is no need to mess with the genome, which may help with eventual applications in humans. | |
| There are various aspects of their nanosensor that the team is now tackling: they will adapt the design in order to enhance the sensitivity on voltage; they plan to combine the sensor with a biological system to conduct measurements in living cells and possibly simple organisms; and they will explore the targeting of the structure to certain cell types by adapting the DNA origami. | |
| "The next major step forward for us will be to integrate a FRET-based optical voltage sensing mechanism into DNA nanopores that self-insert into lipid membranes," Keyser concludes. "Following our successful proof-of-principle study, we believe that DNA origami based optical voltage sensing devices have great potential to be applied for live-cell imaging of transmembrane potentials in the near future." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
