| Posted: May 19, 2006 | |
Attaching biomolecules to carbon nanotubes |
|
| (Nanowerk Spotlight) Among the many potential biology-related applications proposed for carbon nanotubes (CNTs) are high-sensitivity biosensors and bio-fuel cells. In order to create the synergy between the biomolecules and CNTs required to realize these applications, biomolecules, such as proteins and DNAs, must be connected to the CNTs. A useful, simple and universal method to attach biomolecules onto carbon nanotubes with covalent bonding was developed by researchers at Rensselaer Polytechnic Institute. | |
| Before their paper, titled "Protein immobilization on carbon nanotubes via a two-step process of diimide-activated amidation", published in issue 1, 2004 of Journal of Materials Chemistry not a lot of work was done on attaching proteins to CNTs. In the research that was done, proteins were either noncovalently bonded on CNTs or were attached through a harsh process, so the final connection is either very loose or the survival chance of attached protein is very small. | |
| The work done by the Rensselaer scientists provides a universal approach of forming bio-nano materials which have numerous applications. Kuiyang Jiang, first author of the paper, explained to Nanowerk the three core findings of their research: | |
|
|
|
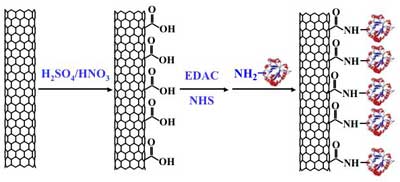 |
Schematic view of diimide-activated attachement process (Source: Rensselaer Polytechnic Institute) |
| The covalently bonding of molecules to the CNTs was done in a two-step process, carried out at room temperature in buffer solutions and was accomplished in a short time, which maximizes the survival rate of biomolecules. | |
| Jiang explains the two-step process: "carboxylic acid groups are first converted to active esters via diimide-activation, and then the active esters are reacted with the amine groups on proteins without the presence of diimide. This two-step process avoids intermolecular conjugation and guarantees the uniform attachment on carbon nanotubes." | |
| Follow-up work to this initial research was done by attaching PAMAM dendrimer onto CNTs through a similar approach as the one described above. This approach provides a universal and efficient method to attach nano-entities with NH2 groups to carbon nanotubes at ambient conditions. | |
 |
TEM Image of PAMAM-MWNT Heterostructure (Source: Rensselaer Polytechnic Institute) |
| This work, titled "Covalent Bonding of Nano-entities with NH2 Groups onto Multi-walled Carbon Nanotubes" was presented at the 2004 MRS Fall meeting in Boston. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
