| Jul 19, 2019 | |
'Diamonds from the sky' chemistry converts greenhouse gas into valuable nanocarbon materials |
|
| (Nanowerk Spotlight) Carbon nano-onions (CNOs), a less studied morphology of carbon nanomaterials, are exotic structures with extraordinary properties and numerous applications. The literature refers to this carbon morphology – nested concentric buckyballs – with various terms such as onion-like structures, carbon nano-onions, carbon onions, carbon spheres, or nested fullerenes. | |
| "Carbon nano-onions have a range of remarkable applications, but these applications have been largely ignored due to their high synthesis cost, " Stuart Licht, a professor of chemistry at George Washington University, tells Nanowerk. "CNO applications often focus on the confined, high surface area of chargeable surfaces or symmetry of their morphology. Examples include ultrahigh power super capacitors with unusually high charge storage; maximum anodic capacity lithium batteries; increased capacity gas, and energy storage materials; increased activity in heterogeneous catalysis; and solid lubricants in the form of ultra strong 'mini ball bearings'." | |
 |
|
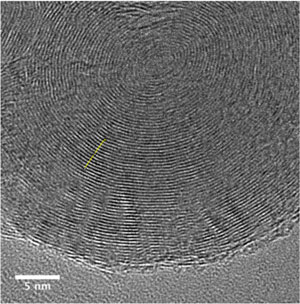
| TEM of a carbon nano-onion. (Image: Licht Group, George Washington University) | |
| So far, the high cost and carbon footprint of producing CNOs – by treatment of commercially available nanodiamonds, by the combustion of naphthalene, or by Chemical Vapor Deposition – has been a considerable constraint on their applications. | |
| We have reported on previous work by Licht's group using molten carbonate chemistry to transform atmospheric carbon dioxide (CO2) into valuable products, for instance carbon nanotubes or carbon nanofibers. | |
| Licht calls this approach diamonds from the sky, referring to atmospheric CO2 being the precursor, carbon being the material that diamonds are made of, and also hinting at the high value of the products. | |
| Processes that transform the greenhouse gas CO2 to a valuable product incentivize its 'mining' and removal, either from industrial flue gas or directly from the air, and could become an important component of climate change mitigating tools. | |
| In new work, reported in Advanced Sustainable Systems ("Carbon Nano-Onions Made Directly from CO2 by Molten Electrolysis for Greenhouse Gas Mitigation"), Licht's team has used their 'diamonds from the sky' chemistry as a high yield pathway to produce inexpensive, stable carbon-nano onions directly from carbon dioxide. The carbon dioxide reactant replaces nano-diamonds as the reactant to form the nano-onions. | |
| In a previous Nanowerk Spotlight, we reported the team's high yield, molten carbonate electrolytic transformation of CO2 to CNTs (referred to as the C2CNT process). In their new study, the researchers provide for the first time an exploration of the initial phases of molten carbonate electrolysis CNT growth, showing their relevance to discovery and introduction of an alternative high yield carbon nano-onion synthesis. | |
| By excluding the CNT transition metal nucleating seeds, the C2CNT process (probably now better called C2CNO) produces a high yield of stable carbon nano-onions. In contrast to previous CNO synthesis methods, CO2 rather than diamond is used the sole precursor. | |
 |
|
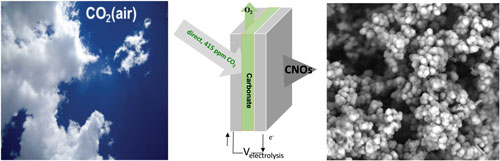
| High yield electrolytic synthesis of carbon nano-onions from CO2, either directly from the air or from smoke stack CO2, in molten carbonate. (Image: Licht Group, George Washington University) | |
| The source of CO2 to produce these CNOs can be the consumption of industrial flue gas or CO2 directly captured from the air. | |
| An economic assessment, detailed in the paper, shows that this new process is highly attractive. "The upper-bound estimate of the new chemistry of CNO production by molten carbonate electrolysis – excluding anode costs to be determined – of $1000 per ton, is several orders of magnitude less than alternatives," Licht points out | |
| The team is now working on realizing larger-scale production of the CNOs to accelerate the process' contribution to climate mitigation efforts, such as in their ongoing C2CNT Carbon XPrize Finals Demonstration Plant. | |
| "Our C2CNT research team is working on bringing to fruition a carbon based economy with change comparable to the introduction of the plastics economy," Licht concludes. "The carbon economy will have common products, such as lubricants, batteries and structural materials replaced by stronger, more functional, carbon products, and provide the basis to remove and the greenhouse gas carbon dioxide from the anthropogenic carbon cycle." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
