| Dec 23, 2019 | |
Bubble- and convection-assisted trapping significantly speed up nanotweezers |
|
| (Nanowerk Spotlight) Plasmonic 'nanotweezers' are structures made of noble metals that concentrate and absorb light, resulting in 'plasmonic hotspots' that make it possible to manipulate nanoscale objects suspended in a fluid. The ability to manipulate small suspended particles such as cells, micro- and nanoparticles plays an essential role in the advancement of areas ranging from nanotechnologies to microbiology and molecular medicine. | |
| Trapping or sensing of nanoparticles using nanotweezers suffers from an intrinsic problem of low throughput as the particle delivery process is often diffusion-limited. In other words, it often takes a long time for particles to diffuse into close proximity – usually within tens of nm – of nanoapertures before they can be trapped or detected due to the short-range plasmon-enhanced electromagnetic fields at the nanoapertures. | |
| The trapping time increases substantially for samples with low concentrations of particles where the diffusion-limited trapping of a single particle at the plasmonic nanoaperture could take as long as several hours. | |
| This limits the application of nanotweezers in various fields, particularly applications requiring large data sets to get statistically relevant data. | |
| Solving this challenge is the prime motivation behind new work conducted at Professor Yuebing Zheng's research group at The University of Texas at Austin and reported in Nano Letters ("Overcoming diffusion-limited trapping in nanoaperture tweezers using opto-thermal-induced flow"). | |
| "We developed bubble- and convection-assisted trapping techniques that exploit Marangoni and Rayleigh-Bénard convection in solution to overcome the diffusion-limited trapping in nanoaperture tweezers and increase their throughput," Dr. Abhay Kotnala, the paper's first author, tells Nanowerk. "Our convection-assisted traps enable a reduction of 1-2 orders of magnitude in particle-trapping time compared to that of a diffusion-limited trap." | |
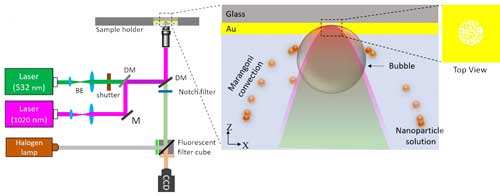 |
|
| Schematic of the experimental set-up for bubble-assisted trapping. The enlarged section shows the AuNIs-encapsulated nanoaperture with the bubble and the induced flow in the solution chamber. DM: Dichroic mirror, BE: Beam expander, M: Mirror, CCD: Charge-coupled device. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| "For example" he elaborates, "at a concentration of 2×107 particles/ml, average particle-trapping times in bubble- and convection-assisted trapping were 7 and 18 seconds, respectively, compared to more than 300 seconds in the diffusion-limited trapping. Trapping of a single particle at an ultralow concentration of 2×106 particles/ml was achieved within 2-3 minutes, which would otherwise take hours with diffusion-limited trapping." | |
| Previously, researchers have proposed electro-thermo-plasmonic effects to solve the challenge of diffusion-limited trapping. However, this has not been effective for single nanoapertures or resulted in ensemble trapping of particles and required the addition of external AC fields. | |
| In contrast, the Zheng group's novel techniques provide a purely optical way to transport particles from 'far-off' distances and trap one particle at a time using a single nanoaperture. | |
| Specifically, the team uses the opto-thermal-induced flow generated from bubble-induced Marangoni and Rayleigh-Bénard convection to rapidly deliver particles from large distances to the nanoaperture tweezers without relying on normal diffusion. | |
| "In addition to increasing the throughput of nanoaperture tweezers, the optothermal-induced flow used in convection- and bubble-assisted traps could also be applied to enhance the performance of nanoaperture-based plasmonic biosensors," Kotnala points out. "The flow created using both techniques could rapidly deliver analytes to the functional surfaces of the sensors and simultaneously increase their local concentration to improve the assay time and sensitivity of the sensor." | |
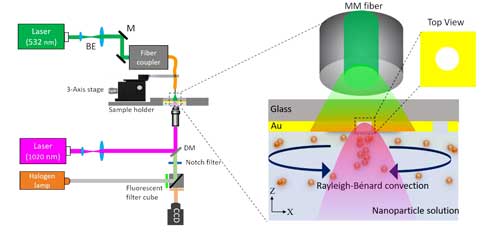 |
|
| Schematic of the experimental set-up for convection-assisted trapping. The enlarged section shows a MM fiber illuminating a large area around a single nanoaperture and the Rayleigh-Bénard convection inducing flow in the solution chamber. DM: Dichroic mirror, BE: Beam expander, M: Mirror, CCD: Charge-coupled device. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| The researchers hope that their techniques will greatly improve the throughput of nanoaperture tweezers systems, which will expand their applications in the study of both synthetic and biological nanoparticles at the single-molecule level. | |
| According to the team, currently both techniques are best suited for particle concentrations in the femtomolar range and above. | |
| Caption: Bubble-assisted trapping of 200 nm fluorescent polystyrene nanoparticles using a single circular nanoaperture. (Video Zheng research group) | |
| "We would like to further optimize our techniques so that they are applicable to particle concentrations in the sub-femtomolar range," says Kotnala. "This will open up the application of nanoaperture tweezers for biological particles found at sub-femtomolar concentrations in their native environment. | |
| "We also plan to extend the application of our techniques to nanoaperture-based plasmonic biosensors where the techniques could be used to improve their assay time and sensitivity," he adds. | |
| Caption: Convection-assisted trapping of 200 nm polystyrene nanoparticles at a concentration of 2x107 particles/mL. (Video Zheng research group) | |
| One of the main challenges that the team faces with their bubble-assisted trapping technique is the immobilization of the particles at the nanoaperture rather than their capture at the bubble-water interface followed by trapping at the nanoaperture, particularly when the laser is turned on for a longer period or at high concentration of particles. | |
| For effective bubble-assisted trapping, it is important to control the laser “ON” time so that most of the particles are only driven and captured at the bubble-water interface. Immobilization of particles on the substrate reduces the effectiveness of the technique and sometimes makes the nanoaperture unusable. | |
| In order to make the techniques more robust and applicable to any concentration of particles, the researchers plan to explore surface modifications of the nanoaperture tweezers that prevent immobilization of particles, independent of laser “ON” time or particle concentrations. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
