| Mar 30, 2020 | |
Manipulating noble metal aerogels by reductant chemistry |
|
| (Nanowerk Spotlight) As we pointed out in our previous Nanowerk Spotlight ("Freeze-thaw made noble metal aerogels: Clean and hierarchical materials for photoelectrocatalysis"), noble metal aerogels are widely investigated in particular for electrocatalysis applications due to the combination of their large specific surface areas and the high catalytic activity of noble metals. | |
| "One of the key issues that is holding back widespread applications of these noble metal aerogels is a lack of understanding of the underlying structure-performance correlations," Dr. Ran Du, a Research Fellow in the Eychmüller Group at Technical University Dresden, tells Nanowerk. "Presumably, this is caused by an insufficient understanding of the sol-gel process that limits manipulating versatile parameters, such as ligament sizes, compositions, and spatial element distributions." | |
| Since noble metal aerogels (NMAs) debuted in 2009, the development of the reductant-directed fabrication method can be considered a milestone. Previously, researchers prepared noble metal hydrogels in two steps by first synthesizing metal nanoparticles with metal salts and reductants, and then using gelation initiators to assemble these nanoparticles into gel networks. | |
| In 2013, scientists found that a particular reductant – sodium borohydride (NaBH4) – can directly transform metal salts into a gel, thus combining the previously necessary two steps into one and yielding clean NMAs without introducing extra initiators. | |
| This strategy has since been widely adopted and became one of the most popular synthesis methods in the field of NMAs. | |
| "However, two crucial issues remain unsolved," Du points out: "First, the fabrication time at room temperature with a classical metal precursor concentration (∼0.2 mM) is extremely long; and second, the underlying mechanism has never been fully explained. Hence, the method is limited to certain metal systems and does not meet the requirements for practical production." | |
| An international team of researchers from TU Dresden, Purdue University, and Wenzhou University joined together to address the above issues. They reported their findings in Nature Communications ("Unveiling reductant chemistry in fabricating noble metal aerogels for superior oxygen evolution and ethanol oxidation"). | |
| By unveiling multiple roles of reductants (i.e., as reducing agents, stabilizers, and initiators) as well as the underlying gelation mechanism via combined experimental and theoretical approaches, the researchers developed an excessive-NaBH4-directed gelation strategy. "Excessive" means that in contrast to previous reports, where the NaBH4-to-metal-salt ratio (R/M) was set to 1.5–5, the team for this work adopted a R/M ratio of 100. | |
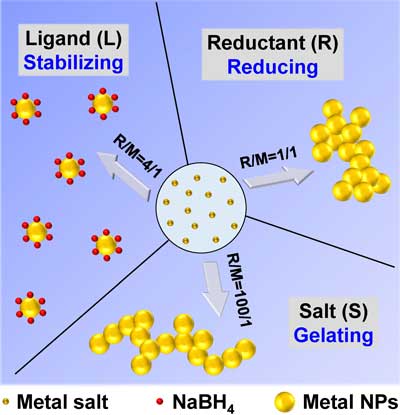 |
|
| Figure 1. Multiple roles of reductants. (Reprinted with permission by Nature Publication Group) | |
| One of the results was that the gel formed within 4–6 hours at room temperature, substantially faster than that of previous NaBH4-triggered gelation systems that took several weeks at this temperature. | |
| Taking advantage of the unprecedented destabilization capacity of this method, the team acquired gold aerogels with a record-high specific surface area (59.8 square meters per gram) by activating the ligand chemistry, and expanding the composition space to all 8 noble metals – gold (Au), silver (Ag), palladium (Pd), platinum (Pt), ruthenium (Ru), rhodium (Rh), iridium (Ir), and osmium (Os) – thus discovering new phenomena (i.e., spontaneous combustion) and obtaining various high-performance electrocatalysts for the ethanol oxidation reaction (EOR) and oxygen evolution reaction (OER). | |
| The team's in-depth study identified the possible roles played by NaBH4 and the underlying mechanisms of the reductant-directed gelation process. | |
| First, broad electrolyte-type reductants, which can dissociate in water and release ions, were shown to directly destabilize metal salts to form the corresponding gels. Three roles of reductants appear in sequence with increasing reductant-to-metal ratio (R/M): the reducing agent, the ligand, and the salting-out agent. | |
| At a low R/M (<2), metal salts are reduced to form nanoparticles, and the unprotected nanoparticles can merge together to form gels driven by their large surface energy. At a medium R/M (2-50), anions from reductants can serve as ligands to stabilize the as-formed nanoparticles, giving rise to stable nanoparticle solutions. In the high R/M region (≥50), a large amount of NaBH4 can destabilize the nanoparticle solution by a salting-out effect. | |
| Second, in conjunction with previous work from the same group (Science Advances, "Specific ion effects directed noble metal aerogels: Versatile manipulation for electrocatalysis and beyond"), the synergy of cations and anions in a salt for triggering gelation was disclosed. | |
| Third, NaBH4 can serve as an extremely powerful reductant for creating gels owing to the uniqueness of BH4- anions. Using gold as an example, the scientists showed that BH4- features a strong binding energy with gold nanoparticle surfaces, while concurrently decomposing to BO2- and hydrogen on the nanoparticles' surface. | |
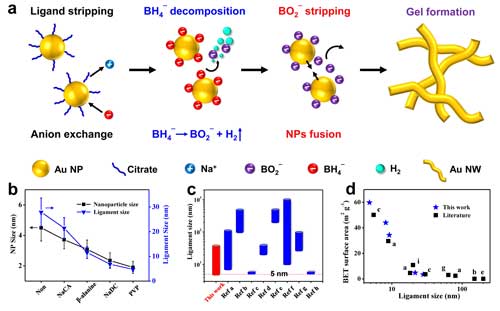 |
|
| Figure 2. (a) The underlying mechanisms of excessive-NaBH4-directed gelation method. (b) Modulation of the ligament size by ligand chemistry, and (c-d) comparison of ligament size and specific surface areas of gold aerogels from this work and literature. (Reprinted with permission by Nature Publication Group) (click on image to enlarge) | |
| "Because BO2- has a very low binding energy with nanoparticles, while hydrogen can propel the movement of nanoparticles, this in situ reaction can substantially promote the collision and fusion of nanoparticles to form gel networks," explains Du. | |
| He adds that they were able to optimize various important electrocatalysis processes. For example – and counterintuitive to the assumption that surfactants will deactivate catalysts – they found an unconventional ligand-enhancing effect to boost the electrocatalytic EOR performance of the Au-Pd aerogel by 50%, displaying a forward current density of up to 7.7 times higher than that of commercial Pd/C. | |
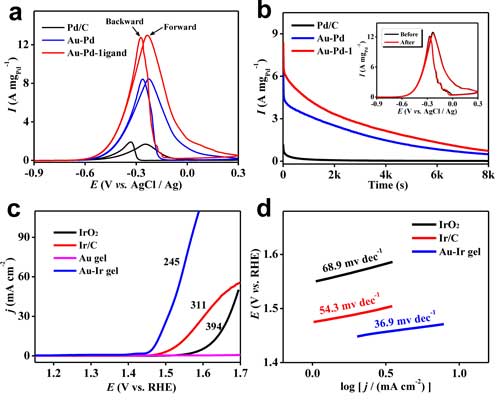 |
|
| Figure 3. (a-b) Ethanol oxidation reaction performance and (c-d) oxygen evolution reaction performance of as-prepared noble metal aerogels and commercial noble metal catalysts. (Reprinted with permission by Nature Publication Group) (click on image to enlarge) | |
| Additionally, the team identified Au-Ir core-shell-structured aerogels as excellent OER electrocatalysts, outperforming commercial Ir/C and IrO2 catalysts in both alkaline and acid environment. | |
| "Our current work not only takes a big step towards the detailed understanding of reductant chemistry and gelation mechanisms, manipulating the microstructures, and enriching the compositions of NMAs, but also opens a new dimension for devising high-performance electrocatalysts by taking advantage of the ligand effects," Du concludes. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
