| May 04, 2020 | |
Sintering advanced ceramics membranes for safe solid-state batteries in just 10 seconds |
|
| (Nanowerk Spotlight) Ceramics are an important class of materials with widespread applications in electronics and energy storage due to their high thermal, mechanical, and chemical stability. Sintering is a critical processing technology in the production of ceramic materials that uses high heat to compact ceramic powders into a solid form. Reducing the porosity of ceramics improves their mechanical properties. | |
| Sintering of pure oxide ceramics requires relatively long processing times (about 20 hours) and high temperatures of 800 degrees Celsius or more. | |
| The development of modern materials such as high-performance ceramics often relies on computational predictions as a valuable tool in accelerating the discovery process. It is essential to experimentally confirm the material properties of such predictions. | |
| "Unfortunately, the screening rates of predicted materials are limited by the long processing times and poor compositional control from volatile element loss in conventional ceramic sintering techniques," Liangbing Hu, the Minta Martin Professor in the Department of Materials Science and Engineering at the University of Maryland, tells Nanowerk. "For instance, during our solid state battery research, we encountered the massive challenge posed by conventional sintering methods on fabricating solid state electrolytes due to the prolonged sintering process, severe volatile element loss, high porosity, and poor composition integration." | |
| He points out that the poor property of the solid state electrolytes (SSEs) results in the inferior electrochemical performance of the battery. To overcome these limitations, Hu's team developed an ultrafast high-temperature sintering (UHS) technique to fabricate SSEs with dense structure and excellent electrochemical performance. | |
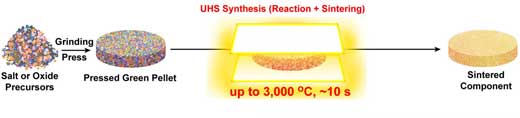
| To conduct the process, the researchers sandwich a pressed green pellet of ceramic precursor powders between two Joule-heating carbon strips that rapidly heat the pellet through radiation and conduction to form a uniform high-temperature environment for quick synthesis (solid-state reaction) and reactive sintering. In an inert atmosphere, these carbon heating elements can provide a temperature of up to 3000 °C, which is sufficient to synthesize and sinter virtually any ceramic material. | |
| Comparing their new synthesis method with key parameters of conventional rapid sintering methods, the UHS technique has a much higher heating rate (~103–104 °C/min) and cooling rate (up to 104 °C/min); requires much less sintering time (1–10 seconds); can reach a temperature of up to 3000 °C, and features minimal sample requirements, making it a general sintering method. Additionally, the UHS technique enables the realization of 3D printing structures and co-sintering of multilayer structures. | |
 |
|
| Schematic of the ultrafast high-temperature synthesis process, in which the pressed green pellet of precursors is directly sintered into a dense ceramic component at a high sintering temperature of up to 3000 °C in ∼10 seconds. (© Science) | |
| The team reported their findings in Science ("A General Method to Synthesize and Sinter Bulk Ceramics in Seconds") | |
| "By optimizing the sintering process, UHS becomes a universal method to accelerate the fabrication of high-temperature ceramic materials," notes Dr. Chengwei Wang, the paper's first author. "UHS can combine with computational prediction models and 3D-printing techniques to accelerate the development of functional materials and structural design." | |
| "The most exciting result in this paper is that we can sinter ceramics in 10 seconds, which is almost 10 000 times faster than the conventional sintering process (∼20 hours)," he adds. | |
| Benefitting from this ultrafast sintering process, the interfacial diffusion and volatile element loss (Li, Na, K et.al.) can be effectively inhibited, resulting in the excellent electrochemical performance of solid-state batteries that use the resulting ceramic membranes. | |
 |
|
| The sintering process of ceramic pellet at room temperature (RT), 1000 °C, and 2000 °C. (Image: Hu Group, University of Maryland) | |
| "Due to the precise control of the element content, its extremely high temperature, element diffusion, and integrated structure, UHS can be applied in multilayer structures design, 3D-printing materials, composite structure fabrication, scalable roll-to-roll of ceramics, and non-oxide high-temperature materials," Hu points out. | |
| In the next stages of their investigations, the team will use the UHS method to fabricate non-oxide high-temperature materials including metals, carbides, borides, nitrides, and silicides. | |
| UHS might also be used to fabricate functionally-graded materials (beyond the simple multilayers demonstrated in the present work) with minimum undesirable interdiffusion. | |
| Also, the ultrafast, far-from-equilibrium nature of the UHS process may produce materials with non-equilibrium concentrations of point defects, dislocations, and other defects or metastable phases, leading to desirable properties. | |
| Finally, this UHS method allows a controllable and tunable temperature profile to enable control of sintering and microstructural evolution. | |
 |
|
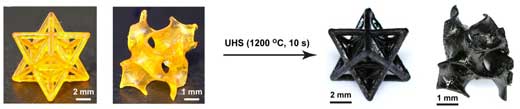
| Left: Photographs of the SiOC polymer precursor 3D-printed as a single material. Right: Photographs of the SiOC samples sintered by the UHS method, showing the uniform material shrinkage and maintained structures. (© Science) | |
| "I think that ultrafast high-temperature sintering will be the most promising high-temperature materials fabrication method in the future," Hu concludes. "The particular challenge for us now is to enlarge the size of the UHS setup to fabricate many ceramics in as fast as 10 seconds. | |
| This high-temperature technology is being commercialized through a University of Maryland spin-off company, HighT-Tech LLC that Prof. Hu co-founded in 2019. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
