| May 11, 2020 | |
Glioblastoma-coated nanotubes target and kill brain cancer cells |
|
| (Nanowerk Spotlight) Research in cancer therapy is putting a lot of effort not only in the development of new drugs, but also in proposing new targeting strategies to selectively treat cancer cells. This would limit the dose of administered drugs as much as possible, decreasing the undesirable effects of chemotherapy, and, at the same time, increasing the efficacy of the treatment. | |
| Glioblastoma multiforme is an extremely aggressive brain cancer characterized by a remarkably high genetic heterogeneity. The word 'multiforme' itself literally means that this tumor can be found in multiple forms (i.e., subtypes). Tumors of different patients display significant histologic and molecular differences. For this reason, finding single molecular markers expressed in all glioblastoma tumors, but not in healthy cells, is particularly difficult. | |
| Consequently, it is extremely hard, and maybe impossible, to find a single molecule that can be exploited to target nanotherapeutics in different glioblastoma subtypes; and if an anticancer therapy cannot selectively target cancer cells, significant damage to healthy cells and negative side effects may occur. | |
| Cancer cell self-recognition is the mechanism by which cancer cells preferentially interact each other through cell adhesion molecules with homotypic binding properties. Recently, this mechanism has been exploited by several research groups in the nanomedicine field to target nanomaterials and drugs to cancer cells. Coating nanoparticles with cell membranes has been confirmed as a winning strategy for targeting particles using homotypic recognition, opening important perspectives in precise and personalized anticancer treatments. | |
| Specifically, the plasma membranes of a specific cancer cell type can be extracted and used to functionalize (e.g., to coat) a drug-loaded nanomaterial. In this case, when the nanotherapeutic comes in contact with the cancer cells, the adhesion molecules on its surface recognize and bind to the adhesion molecules exposed by the cancer cells. Thanks to this phenomenon, the functionalized nanotherapeutic binds cancer cells with remarkably higher efficacy compared to healthy cells. | |
| Solutions that exploit this cellular self-recognition process do not consist of a single marker to be targeted, but in a multitude of homotypic cell adhesion molecules expressed by the cancer cells themselves. | |
| In new work reported in Materials and Design ("Homotypic targeting and drug delivery in glioblastoma cells through cell membrane-coated boron nitride nanotubes"), researchers in Italy exploited for the first time this recognition approach for targeting glioblastoma cells with boron nitride nanotubes (BNNTs), a biocompatible, yet inorganic, nanomaterial. | |
| The team's targeting approach exploits the ability of cancer cells to recognize each other due to similarities present on their membrane that make them different from healthy cells. | |
| "We loaded the BNNTs with doxorubicin, a powerful chemotherapy drug, and then functionalized them with glioblastoma cell membranes," Gianni Ciofani, Research Director of the Smart Bio-Interfaces group at the Italian Institute of Technology, explains to Nanowerk. "Taking advantage of a multi-chamber fluidic bioreactor, we demonstrated that the chemotherapy agent can be specifically delivered to glioblastoma cells for their selective apoptosis (i.e., programmed cell death)." | |
| "Most importantly, although doxorubicin is a particularly aggressive chemotherapy drug, we found no toxic effect in healthy cells, demonstrating the high selectivity of our treatment," he points out. "In this work we also identified the main cell adhesion molecules involved in the process of self-recognition between glioblastoma cells." | |
| Glioblastoma multiforme is one of the most aggressive types of brain cancer, characterized by an extremely high mortality rate. Despite massive research efforts to develop diagnostic and therapeutic tools, the 5-year survival rate is about 5%. Even a small improvement of the treatment efficacy and a decrease of its side effects could provide a considerable improvement of the quality of the patient lives. | |
| For these reasons, the development of innovative approaches allowing for improved selectivity of anti-cancer treatments represents one of the greatest challenges in the oncology field. This work has been carried out in the framework of the SLaMM project funded by the European Research Council (ERC) under the European Union's Horizon 2020 research and innovation program (grant agreement N?709613). This project is focused on the development of drug-loaded nanomaterials able to selectively target and kill glioblastoma cells. | |
| "Our platform acts like a 'magic bullet', which hits the enemy effectively, leaving the 'innocent' unharmed," say Mr. De Pasquale and Dr. Marino, first co-authors of the study. "We subjected our nanovector to several tests to ascertain its degree of selectivity and the results have shown amazing effects. In fact, the nanovector is able to penetrate inside glioblastoma cells avoiding all the other cell lines tested, and once inside cancer cells, it is able to induce cell death by releasing the chemotherapy drug." | |
| In addition, with this system it has been possible to carry doxorubin across a model of the blood-brain barrier. | |
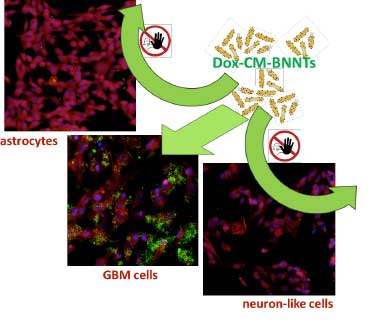 |
|
| Cancer cell membrane-coated boron nitride nanotubes loaded with doxorubicin (Dox-CM-BNNTs) selectively target and kill glioblastoma multiforme (GBM) cells leaving unaffected astrocytes and neuron-like cells. Cell cytoskeleton in red; cell nuclei in blue; Dox-CM-BNNTs in green. (Image: Smart Bio-Interfaces group, Italian Institute of Technology) | |
| This work is currently limited to in vitro studies, although these studies have been carried out on rigorous in vitro complex models resembling the pathological and physiological milieu of glioblastoma, like multi-chambers fluidic bioreactors sustaining co-cultures. | |
| Given the encouraging results of the experiments on this new nanovector, the next step for the research team will be to check its efficacy on in vivo animal models, and, ultimately, be tested on patients with glioblastoma multiforme. | |
| "We could even obtain the external coating of the nanovector directly from the patient's tumor cells, avoiding the problems due to the immune response from which nanomaterials commonly suffer," notes Ciofani. "With this new nanovector, many new pharmacological alternatives to the treatment of glioblastoma multiforme are opening up, widening the spectrum of drugs to be used. A whole series of useful drugs could be re-considered that have been discarded in the past due to their impenetrability through the blood-brain barrier." | |
| "The future of our research and, we suppose, of the research community operating in this field, will be directed to the development of personalized approaches designed for each patient," he concludes. "Our work is directed to this aim and our experimental design suits this purpose. In future, patient biopsies will represent the source of cell membranes to be used for the selective targeting of nanotherapeutics. Recent studies by other research groups have also highlighted that cancer cell membranes are able to provide a vehicle to efficiently cross the blood-brain barrier. This could pave the way for the development of intelligent nanomedicines that, from the bloodstream, selectively accumulate and progressively release anticancer drugs at the site of glioblastoma." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
