| Posted: Jun 09, 2006 | |
A simple method for filtering gold and silver out of water with carbon nanotubes |
|
| (Nanowerk Spotlight) Silver single crystals were facilely synthesized on a large-scale with good reproducibility in water at room temperature in the presence of carboxyl-functionalized carbon nanotubes, without any additional reducing agent/electrochemical reducing, microwave, sonication or irradiations. | |
| Researchers in China and the UK developed an extremely simple "nanocarbon" method to produce Ag/CNT nanohybrids. This process and the resulting nanomaterial could prove very useful for catalysis and chemical biology. Even more, this nanocarbon method can be used to reclaim silver and gold from wastes directly, implying that "nanocarbons touch a water solution and turn it into silver/gold" as Professor Chao Gao remarks. "Hence, it may be quite useful in environmental engineering and relevant areas" he says. | |
| Dr. Gao is first author of a recent paper, titled "Facile and large-scale synthesis and characterization of carbon nanotube/silver nanocrystal nanohybrids" published in the May 26, 2006 online edition of Nanotechnology. Together with colleagues from the College of Chemistry and Chemical Engineering at Shanghai Jiao Tong University in PR China, and the Sussex Nanoscience and Nanotechnology Centre in the UK, he developed a facile and efficient aqueous phase-based strategy to synthesize CNT/silver nanocrystal nanohybrids at room temperature. | |
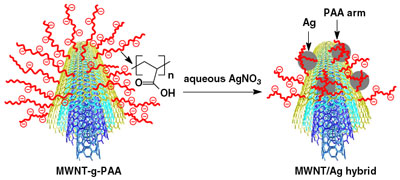 |
Schematic illustration for the generation of MWNT/Ag nanohybrids from anionic nanocatcher (polyacrylic acid-grafted MWNTs, MWNT-g-PAA) and silver nitrate (AgNO3) aqueous solution. (Reprinted with permission from the Institute of Physics Publishing). |
|
Gao explains how their research produced several new findings:
|
|
| Detailed experiments showed that this strategy can also be applied to different CNTs, including SWNTs, DWNTs, MWNTs and polymer-functionalized CNTs. Gao explains that the nanoparticle sizes can be controlled from 2 nm to 10–20 nm and the amount of metal deposited on CNT surfaces can be as high as 82 wt%. | |
| "Furthermore" Gao says, "large-scale (10 g or more) CNT/Ag nanohybrids can be prepared via this approach without the decrease of efficiency and quality." This approach can also be extended to prepare Au single crystals by CNTs. | |
| Based on the merits of this approach, CNT/Ag nanohybrids can now easily be made available on a large scale with good controllability and reproducibility, which paves the way for a wide range of practical applications of such nano-objects in the areas of catalysis, environmental engineering, and chemical biology. | |
| Building on this current work, Gao says they are trying other types of nanocarbon to produce noble metal crystals, exploring the mechanism of this method, and applying the products in catalysis and other fields. They already prepared quantum dots/CNT hybrids ("Multiamino-functionalized carbon nanotubes and their applications in loading quantum dots and magnetic nanoparticles") and magnetic CNTs and used the magnetic CNTs to manipulate cells in a magnetic field ("Magnetic Carbon Nanotubes: Synthesis by Electrostatic Self-Assembly Approach and Application in Biomanipulations"). | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
