| Sep 23, 2020 | |
Turning tiny algae into living micromotors controlled by light (w/video) |
|
| (Nanowerk Spotlight) The structure and function of living systems, for instance bacterial propulsion, are a frequent inspiration for researchers when designing and engineering micro- and nanoscale motors, actuators, sensors, and robots. These platforms translate biological principles into man-made designs, for instance microrobotic systems, and bridge the divide between synthetic machines and the biological world. | |
| Micromotors are tiny engines that convert stimuli, such as chemical fuel, light, magnetic fields or sound, into motion to perform certain tasks. Most of these systems are biohybrids, meaning that they are biological materials that depend on the inclusion of synthetic materials. An example is the use of magnetic nanoparticles to make a bio-based micro- or nanomotor responsive to a magnetic source for actuation and locomotion control. | |
| In new work reported in Nano Letters ("Optically Controlled Living Micromotors for the Manipulation and Disruption of Biological Targets"), researchers from the Institute of Nanophotonics at Jinan University in Guangzhou, China, demonstrate the creation of a living micromotor with multifunctional capability. The distinctive feature of this device is that it doesn't require synthetic materials. | |
| "Because they don't require synthetic materials, our living micromotors are highly biocompatible and sustainable, without the problems of energy consumption and waste production generated during material synthesis of current technologies," Hongbao Xin, a professor in the Institute of Nanophotonics at Jinan University, tells Nanowerk. "Our work represents a step forward in the development of biocompatible, intelligent, and sustainable microrobotic systems with multifunctional capabilities." | |
| Using microalgal Chlamydomonas reinhardtii (CR) cells as the sole material, the scientists made living and multifunctional micromotors controlled by optical force, i.e. light. This system might be particularly useful for indirect manipulation and controllable disruption of biological targets. | |
 |
|
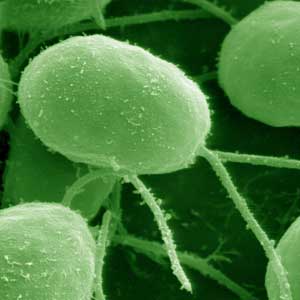
| A false-colored scanning electron microscope image of Chlamydomonas reinhardtii. These tiny algae range in size from 3 to 10 microns. (Image: Louisa Howard, Dartmouth College) | |
| While CR – a unicellular flagellate – has previously been used for micromotor applications, this required the integration of synthetic materials (magnetic particles) onto the cell surface for controlled navigation via magnetic auction. | |
| "Natural CR cells exhibit motility and locomotion capability in their living environment, which motivated us to conduct this work" explains Xin. "This natural locomotion, however, tends to be uncontrollable. By using an optical force via scanning optical tweezers to control the motion, we turned a random and uncontrollable swimming CR cell into a controllable rotary micromotor." | |
| The team demonstrated the controlled rotation and directed locomotion feature of these micromotors in different biological media, including saliva, serum, plasma, blood, and bone marrow fluid. | |
 |
|
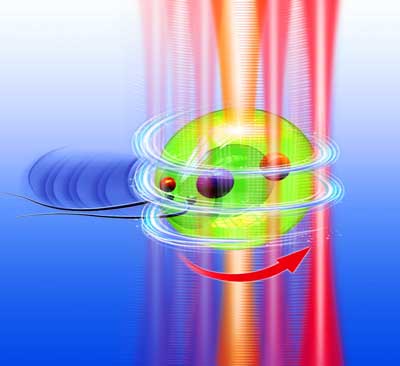
| Turning the natural motility of a Chlamydomonas reinhardtii into a rotational living micromotor via scanning optical tweezers. In this illustration the central light beam shows the optical trap for CR trapping, whereas the gradually varied beams on the cell boundary and the curved red arrow show the annular scanning trap to control the rotation of the motor. (Image: Institute of Nanophotonics, Jinan University) | |
| Many of the reported micromotors exhibit only single functions. In contrast, this CR micromotor can perform multiple functions. For example, the micromotor can be used as an active actuation platform for indirect manipulation/circulation of different targets from immotile particles and cells to motile bacteria. It can also be used for targeted delivery and controlled release of different biological targets. | |
| The rotation and locomotion of the living micromotor can be controlled by adjusting the annular scanning trap and the central trap, respectively. The rotation direction of the motor can be changed by changing the direction of the scanning trap, while the rotation speed can be controlled by the optical power of the scanning trap. This controllable rotation is very useful when the motor is used for performing localized task in different environments, including microfluidics, where targeted delivery and on-demand actuation are necessary. | |
| In particular, the flagella of the micro algae can act as robotic arms with controlled back-and-forth beating on targets during the motor rotation, enabling the disruption and subsequent removal of biological aggregates including in vitro blood clots. | |
| Furthermore, as the researchers point out, the living micromotor can be extended to motor arrays with high reconfigurability and controllability. These motor arrays can work independently and collaboratively with higher efficiency than a single motor, serving as a high-efficiency operation platform to perform different tasks. | |
 |
|
| Illustration of micropumping and indirect manipulation/circulation of different biological targets via the rotational micromotor. (Image: Institute of Nanophotonics, Jinan University) | |
| "Our living micromotor provides many possibilities for biomedical applications and it represents a step forward in the development of biocompatible, intelligent, and sustainable microrobotic systems with multifunctional capabilities," Xin concludes. "As such it falls within the two broad future directions of the micromotor and microrobotic research field: One is in vivo applications, where micromotors and microrobots are designed to perform tasks within an in vivo environment such as targeted drug delivery, thrombi removal, and precise microsurgery. The other one is the application in environmental science, such as environmental remediation, biofilm removal, detoxification, etc." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
