| Feb 16, 2021 | |
How to power implantable and ingestible electronics |
|
| (Nanowerk Spotlight) Imagine edible medical devices that can be safely ingested by patients, perform a test or release a drug, and then transmit feedback to your smartphone; or an ingestible, Jell-O-like pill that monitors the stomach for up to a month. | |
| Devices like these, as well as a wide range of implantable biomedical electronic devices such as pacemakers, neurostimulators, subdermal blood sensors, capsule endoscopes, and drug pumps, can be useful tools for detecting physiological and pathophysiological signals, and providing treatments performed inside the body. | |
| Advances in wireless communication enable medical devices to be untethered when in the human body. Advances in minimally invasive or semi-invasive surgical implantation procedures have enabled biomedical devices to be implanted in locations where clinically important biomarkers and physiological signals can be detected; it has also enabled direct administration of medication or treatment to a target location. | |
 |
|
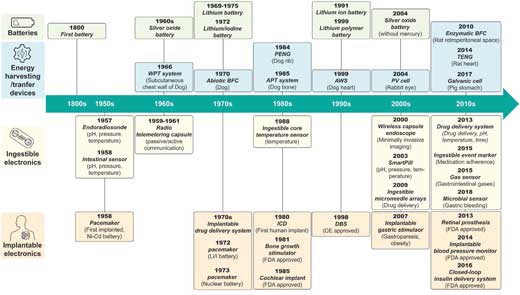
| Timeline of major milestones for implantable and ingestible electronic devices and technology for powering such devices. Listed are the years when batteries suitable to power biomedical devices were first commercialized, in vivo experiments of energy harvesting and transfer devices first occurred, ingestible electronics first appeared, and implantable electronics first appeared. (WPT: wireless power transfer, BFC: biofuel cell, PENG: piezoelectric nanogenerator, APT: acoustic power transfer, AWS: automatic wristwatch system, PV: photovoltaic, TENG: triboelectric nanogenerator). (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
| However, one major challenge in the development of these devices is the limited lifetime of their power sources. The energy requirements of biomedical electronic devices are highly dependent on their application and the complexity of the required electrical systems. | |
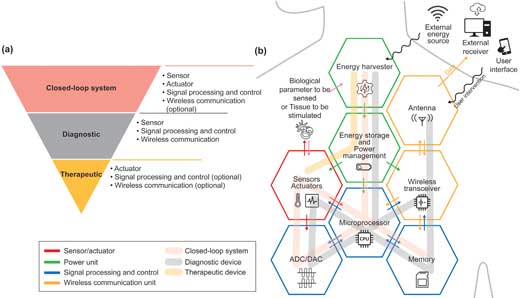
| Biomedical electronic devices can be divided into three main categories depending on their application: diagnostic, therapeutic, and closed-loop systems. Each category has a different degree of complexity in the electronic system. | |
| A review article in Advanced Functional Materials ("Powering Implantable and Ingestible Electronics") discusses these issues in detail and also addresses limitation and challenges. | |
| As illustrated in the figure below, most biomedical electronic devices are composed of a common set of components, including a power unit, sensors, actuators, a signal processing and control unit, and a data storage unit. Implantable and ingestible devices that require a great deal of data manipulation or large quantities of data logging also need to be wirelessly connected to an external device so that data can be transmitted to an external receiver and signal processing, data storage, and display can be performed more efficiently. | |
| The power unit, which is composed of one or more energy sources – batteries, energy-harvesting, and energy transfer – as well as power management circuits, supplies electrical energy to the whole system. | |
 |
|
| a) The system configuration and b) the schematic of functional units of closed-loop, diagnostic, and therapeutic implantable/ingestible electronics. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
Batteries |
|
| Implantable medical devices such as cardiac pacemakers, neurostimulators and drug delivery devices are major medical tools to support life activity and provide new therapeutic strategies. Most such devices are powered by lithium batteries whose service life is as low as 10 years. Hence, many patients must undergo a major surgery to check the battery performance and replace the batteries as necessary. | |
| In the last few decades, new battery technology has led to increases in the performance, reliability, and lifetime of batteries. However, challenges remain, especially in terms of volumetric energy density and safety. | |
| Electronic miniaturization allows more functionalities to be added to devices, which increases power requirements. Recently, new material-based battery systems have been developed with higher energy densities. | |
| Also, battery components can be arranged in different geometric orientations in order to efficiently leverage the limited space in biomedical devices. Researchers have also been focused on developing electrolyte and electrode materials that are nonflammable, nontoxic, and biodegradable in order to improve the safety of batteries. | |
| The review looks at the major challenges of battery development for biomedical applications and discusses solid-state batteries, non-Li metal based batteries, transient batteries, and flexible microbatteries an microsupercapacitors. | |
Energy Harvesting |
|
| Different locations and organ systems in the human body have access to different types of energy sources, such as mechanical, chemical, and electromagnetic energies. | |
| Nanogenerators are innovative self-powered energy harvesters that convert kinetic energy created from vibrational and mechanical sources into electrical power, removing the need for external circuits or batteries for electronic devices (read more in our previous Nanowerk Spotlight: "Nanotechnology for self-powered systems"). | |
| Of particular interest for researchers working on in-body electronics are piezoelectric material-based energy harvesters, or piezoelectric nanogenerators (PENGs), that can convert the mechanical energy present in small vibrations into electrical energy. | |
| PENGs can be worn externally or implanted in the body; they can be used to convert small mechanical vibrations generated by the human body from activities such as walking, breathing, or fluxes in biofluids, into energy to power implantable medical devices. | |
| Another type of nanogenerator are triboelectric energy harvesters. In triboelectric devices, electrostatic charges are generated when two different materials, which have electrically charged surfaces, are brought into contact. A typical triboelectric nanogenerator (TENG) consists of two thin films with opposite tribo-polarity; each film has an electrode attached to its back side. When the materials come into close contact, charges are transferred between the films leaving one side positive and the other negative; when the materials are separated, the transferred charges create a triboelectric potential (read more in our previous Nanowerk Spotlight: "Triboelectric nanogenerators for next-generation wearable health monitoring"). | |
| In addition to these mechanical energy harvesting methods there are chemical energy sources that can be harvested. Examples are galvanic cells and biofuel cells. Both are discussed in greater detail in the review. | |
Energy Transfer |
|
| Energy transfer technologies can deliver energy from outside the body to implanted or ingested devices. If devices are implanted at the locations where there are no accessible endogenous energies, exogenous energies in the form of ultrasonic or electromagnetic waves can penetrate through the biological barriers and wirelessly deliver the energies to the devices. | |
| The types of methods discussed by the authors include mechanical methods such as acoustic power transfer (also called acoustic wireless powering or ultrasonic-based wireless power transmission) and electromagnetic energy transfer, including optical transfer via the photovoltaic effect. | |
| Concluding their review, the authors note that low energy conversion efficiency and power output are the fundamental bottlenecks of energy harvesting and transfer devices. They suggest that additional studies are needed to improve the power output of energy harvesting and transfer devices so that they can be used to power various biomedical electronics. | |
| Furthermore, durability studies of promising energy harvesters should be performed to evaluate their use in long-term applications. For degradable energy harvesting devices, such as friction-based energy harvesters and galvanic cells, improving the device lifetime is essential for use in real-life applications. | |
| Finally, manufacturing cost is another factor to consider when commercializing novel batteries, energy harvesters, or energy transfer devices as power sources for medical devices. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
