| May 03, 2021 | |
Natural silk-like hierarchical fibers created by bioinspired self-assembly |
|
| (Nanowerk Spotlight) Protein fibers such as collagen fibers, hairs, and silks, are fundamental building blocks of life. They play an essential role in motility, elasticity, scaffolding, stabilization and the protection of cells, tissues and organisms. | |
| Artificially creating the hierarchy of these natural protein fibers is crucial to providing strategies for developing advanced fibrous materials, for instance in regenerative medicine. However, this has proven to be very challenging due to the complexity of this biomaterial. | |
| Inspired by the silk spinning of spiders, researchers at the Institute for Frontier Materials at Deakin University have developed a facile strategy to artificially create natural, silk-like hierarchical fibers. | |
| Reporting their findings in Nature Communications ("Bioactive hierarchical silk fibers created by bioinspired self-assembly"), they demonstrate the creation of biomimetic and highly bioactive materials by artificially manipulating the self-assembly of silk protein molecules. | |
| "Our work contributes to the understanding of the natural silk spinning process of spiders and provides a strategy for the design and development of advanced fibrous biomaterials for various applications," Dr. Linpeng Fan, first author of the paper, tells Nanowerk. | |
| Natural materials like the above-mentioned collagen fibers, hairs, and silks have evolved for thousands of years. Their unique hierarchical structure, properties and functions are very intriguing, which is motivating researchers to artificially create biomimetic advanced functional materials. | |
| Natural protein fibers usually have a common structure with bundles of smaller fibrils in their long axis. One single silk fiber, for instance, is a bundle of thinner fibrils aligned along its long axis where the width of the fibrils ranges from nanometers to sub-micrometers. | |
| Currently, it is very challenging to artificially duplicate the hierarchy of natural silk fibers – including silkworm silk fibers and spider silk fibers – by artificially manipulating the self-assembly of silk protein molecules. | |
| Inspired by the silk spinning of spiders, Fan and his collaborators have developed a facile strategy to artificially create natural silk-like hierarchical fibers and demonstrate their high bioactivity. | |
 |
|
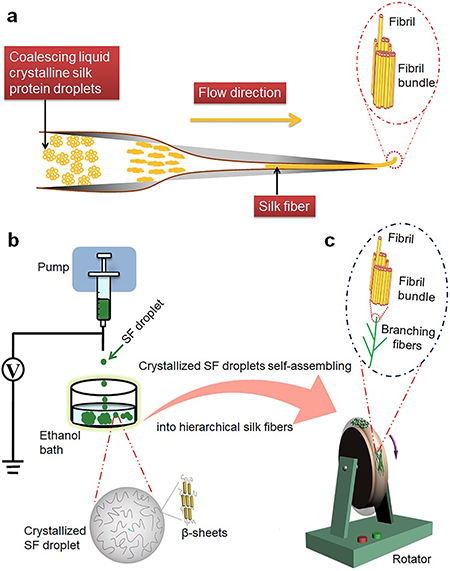
| Figure 1. Schematic illustration of crystallized droplets of silk protein molecules self-assembling to hierarchical suprafibrillar silk fibers. a) Schematic mechanism illustration of the liquid crystalline model for formation of in vivo silk fibers. b) Formation of crystallized droplets from silk fibroin (SF) molecules. c) Crystallized SF droplets self-assembling to hierarchical suprafibrillar silk fibers. (Reprinted with permission from Nature Communications) | |
| The first step of the team's strategy is to fabricate crystallized droplets by electrospraying droplets of aqueous silk fibroin (SF) solution into an ethanol bath in which the amorphous structure of SF with dominant random coils is induced to a β-sheet-rich crystalline structure (Fig. 1b). Subsequently, the crystallized droplets are induced to self-assemble by a circular rotary system (Fig. 1c). | |
| "Differing from the spinning system in which the fibers are extruded instantly via the spinneret, in our technique, the circular assembly system focuses on the self-assembly of crystallized SF droplets to form hierarchical silk fibers," Fan explains. "Compared to the spinning process, one great advantage of the circular assembly system is that it allows us to easily monitor the entire assembly process of silk fibers by investigating the resultant structures at different assembly stages, such as different time points of the self-assembly process." | |
| Furthermore, he points out, the self-assembly process can be programmed by adjusting the assembly parameters to produce branched hierarchical suprafibrillar silk fibers at the micrometer and even nanometer scale. | |
| Based on this work, the team is currently developing bioactive and smart biomaterials for use in the regenerative medicine and health monitoring. | |
| "The future focus in my research field is on the development of bioactive and smart biomaterials for various applications by emulating the structure, properties and functions of materials in nature – a task that provides my colleagues and me with plenty of challenges," Fan concludes. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
