| Posted: May 22, 2008 | |
Understanding and measuring miniscule forces at the molecular level |
|
| (Nanowerk Spotlight) 'Reverse engineering' is the process of discovering the technological principles of a device or system through analysis of its structure, function and operation, often by taking it apart and analyzing its workings in detail. This approach is a common practice among industrial companies who use it to analyze the competition's products, be it cars or MP3 players, to understand where the latest product improvements come from and how individual components are made. | |
| An increasing number of scientists apply a similar approach to nature's own 'micro- and nanotechnology' systems. They believe that learning from nature's designs and engineering successes is more likely to provide the cues for designing practical nanodevices than by simply applying a 'trial and error' approach. The basic idea is that natural materials and systems can be adopted for human use beyond their original purpose in nature. Popular examples of this approach are DNA computing and DNA 'nanomachines'. | |
| While you won't be seeing any functional DNA computers sitting on your desktop for quite a while yet, other examples of 'reverse' biophysics work and have already proven quite useful, for instance the use of individual red blood cells as reliable, ultrasensitive mechanotransducers. | |
| Reliable measurement of miniscule forces is increasingly recognized as a very sensitive way to characterize molecular and cellular interactions in biology, medicine, and by the biotechnology and pharmaceutical industry. Changes in cell mechanical properties have been shown to correlate with metastatic potential and stem-cell differentiation. Forces play a dramatic role in drug targeting and delivery. Another example is the adhesion cascade of white blood cells during inflammation. One important lesson learned in recent years is that biochemistry alone cannot predict how the interaction between two reactants is affected by force – and yet most biomolecular interactions are actually stress-bearing in one way or another. | |
| A technique – originally pioneered by Prof. Evan Evans at the University of British Columbia and called 'Biomembrane Force Probe' (BFP) – uses the deformation of a red blood cell under tension as a force sensor ("Energy landscapes of receptor–ligand bonds explored with dynamic force spectroscopy"). | |
| A new paper puts forward a rigorous indepth study of this – one of the earliest – working examples of adopting a biomaterial to a useful task beyond its natural function. Published in the April 16, 2008 online edition of Applied Physics Letters, it spans an arc from theoretical membrane physics and numerical analysis, over innovative instrument development and advanced experimentation, to the eventual adaptation of a well-characterized biological object as a device capable of high-resolution nanomechanical measurements ("Biophysics in reverse: Using blood cells to accurately calibrate force-microscopy cantilevers"). | |
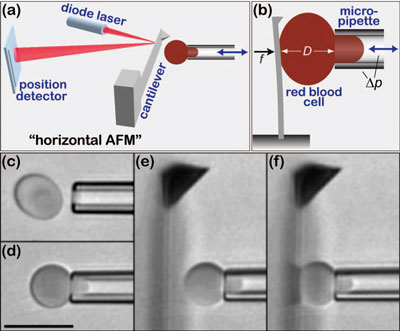 |
|
| Overview of the experimental setup and procedure. [(a) and (b)] The core components of the horizontal AFM are arranged perpendicular to the optical axis of an inverted microscope, providing a side view of the cantilever and allowing for easy integration of a micropipette setup. The double arrow denotes pipette translation to/from the cantilever by closed loop piezoactuation. (The sketches are not to scale.) [(c) - (f)] Video micrographs (bar=10 µm) show how a pre-swollen (at ∼150 mOsm) red blood cell is first picked up in a pipette cylindrical tip, ID ∼2.2 µm [(c) and (d)]), then manipulated close to the tip of the cantilever (e), and repeatedly pushed against the flat of the cantilever (f). (The side view of the ∼30-µm-wide cantilever creates a blurry diffraction pattern; only the cantilever tip appears more or less in focus.) (Reprinted with permission from American Institute of Physics) | |
| "Examining how a red blood cell can be used as a force transducer, and could be applied to measure forces between individual molecules and cells or between molecules, we verified the viability of using pipette-aspirated red blood cells as ultrasensitive force-measuring devices" Dr. Volkmar Heinrich explains to Nanowerk. "In conjunction with our custom-built, cantilever-based 'horizontal AFM', we were able to vary critical parameters such as the aspiration pressure and the position of cell-cantilever contact. Our measurements yielded excellent agreement with our theoretical predictions, proving that among other applications, this red blood cell transducer can be used to reliably calibrate the spring constants of AFM cantilevers." | |
| Heinrich, assistant professor in the Department of Biomedical Engineering at UC Davis, and Chawin Ounkomol, a graduate student in his group, address the specific case of the BFP transducer's precision in their paper. Their work has tackled two issues simultaneously: It refines BFP and similar, red-cell-based force instruments, and it also presented a new type of cantilever-based force probe that can be viewed as a combination of a 'horizontal AFM' and an automated micropipette-manipulation system. | |
| "It was this combination that allowed us to directly test the red-cell force transducer against the elastic microlevers used in commercial AFMs" says Heinrich. "Our tests have validated that, in conjunction with the correct theory, red blood cells can indeed be used as reliable mechanical nanodevices. At the same time, we were able to turn the tables of conventional nanobiophysics and technology by demonstrating that a biological object could accurately calibrate a man-made device, in this case, an AFM cantilever. In fact, a careful look at the data reveals that this method of cantilever calibration may well be more precise than common calibration techniques." | |
| Heinrich mentions that his team is viewing this particular calibration as an added bonus to their more fundamental innovations in theory and instrument design. "Of course, it is not the most practical method to calibrate AFM cantilevers, and it will unlikely become a standard technique. That said, I suspect that whenever we will want to calibrate a cantilever with excruciating accuracy in the future in our lab, we might just fall back onto this method." | |
| Heinrich explains that red blood cells exhibit quite impressive mechanical characteristics. They remain in the human blood circulation system for 3-4 months. During this time, they are exposed to continuous stress exerted by the blood stream. Each red blood cell has to squeeze numerous times through capillaries that are narrower than its own largest dimension (when relaxed). It is hard to imagine a synthetic material with the same combination of mechanical integrity and flexibility. Therefore, it is perhaps not surprising that researchers have begun to explore alternative uses of this amazing biomaterial. | |
| "When a red blood cell is 'sucked' onto the tip of a micropipette of the right caliber, its mechanical properties dictate that it forms a spherical part outside the pipette and a small cylindrical 'projection' inside the pipette" says Heinrich. "Similar to a pressurized balloon, the spherical part exhibits a certain amount of 'springiness'. Based on our theoretical work, we are able to precisely predict this springiness – in other words, the relationship between the cell deformation and the force exerted by the cell. Even better, the springiness of the spherical, balloon-like part depends on the suction pressure applied through the pipette. Since we can precisely control this pressure, we can adjust the amount by which the cell deforms when 'squeezed' against another object. Taken together, not only do we know the force that is exerted by the cell on the object, we can even fine-tune it by changing the suction pressure in the pipette." | |
| Skeptics have wondered how well this idea might really work in practice, posing questions like: Are the relevant red-cell physics properly accounted for? Do all red blood cells behave in the same way in this configuration? | |
| The UC Davis team's research soundly answers these questions: "The only requirements for a red blood cell to operate reliably as an ultrasensitive force probe are a fluid membrane and cell interior, reasonable lubrication between the membrane and pipette wall, and at least a moderate aspiration pressure." If these conditions are fulfilled, all else that one needs to know are the value of the aspiration pressure and the overall cell dimensions. The amazing constitution of red blood cells ensures that the rest is routine. | |
| Heinrich points out that it is important to place this work into a larger perspective. Only little more than a decade ago, the possibility that one could directly measure the forces acting between molecules appeared remote. The advent of nanotechnology has dramatically changed this perception. Today there are a number of tools that can be used to characterize the nanomechanics of biomolecular and cellular interactions. Examples are optical tweezers, magnetic pullers, cantilever-based instruments like the atomic force microscope (AFM), and the BFP. | |
| "With new tools came new concepts" he says. "It turns out that the mechanical rules that govern the nanoworld are quite different from our everyday, macroworld experience. New insights taught us that the accurate characterization of the extremely small forces of 'weak' interactions is more demanding than at first thought. We know now that a large dynamic range of forces is required to properly characterize such weak interactions – ideally this range should be broader than what can be achieved with a single instrument. The future of the field of 'nano-interactions' will see an increased demand for very precise force-probe instruments with complementary dynamic ranges." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
