| Jun 10, 2021 | |
Targeting cancer cells over healthy cells with nanoparticle shape |
|
| (Nanowerk Spotlight) Researchers working on nanomedicine drug delivery have known for a few years now that the shape and geometry of nanoparticles can have a significant impact in dampening adverse reactions to nanoparticle injection as well as increase the uptake efficiency and circulation time of nanopharmaceuticals in the body. | |
| Studies have shown that keeping the material, volume, and the targeting antibody the same, a simple change in the shape of the nanoparticle enhances its ability to target specific tissues. In particular rod-shaped nanoparticles – or nanorods – as opposed to spherical nanoparticles, appear to adhere more effectively to the surface of certain cells. | |
| So the obvious next question is if it is possible to specifically target only cancer cells and leave the healthy cells alone. | |
| "The challenge of selective targeting using antibodies was highlighted in 2016 in a review in Nature Reviews Materials, stating that with antibody targeting less that 1% of nanoparticles reached the target of a solid tumor," Professor J. Justin Gooding from the School of Chemistry at the University of New South Wales, tells Nanowerk. "So we launched a program to use particle shape and size to exploit uptake pathways unique to the cancer cells as a targeting mechanism." | |
| What they found was that cancer cells have a preference to take nanorods over nanospheres. This is related to the fact that cancer cells can take up nanoparticles by macropinocytosis, an uptake pathway not available to healthy cells. By using the rod-shaped nanoparticles, in combination with inhibitors to other uptake pathways the researchers achieved targeting the cancer cells over the healthy cells. | |
| The possible benefits from this novel technique are twofold: Restricting healthy cells from taking anticancer drugs to help reduce the side effects of drugs for patients; and helping overcome chemoresistance (the uptake of the chemo drug into healthy cells in the tumor setting is one of the main causes of chemoresistance). | |
 |
|
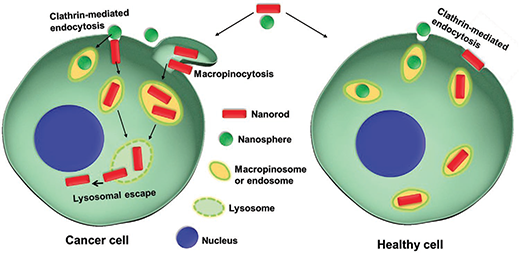
| Schematic illustration of nanoparticle shape selection on cancer and healthy cells. A mixture of nanospheres and nanorods together are exposed to cancer and healthy cells. The cancer cells exhibit a strong preference for the nanorods over the nanospheres, and the nanorods can escape lysosomes in the cancer cells. The healthy cells show a less dramatic preference for the nanorods over the nanospheres. (Reprinted with permission by Wiley-VCH Verlag) | |
| In Advanced Functional Materials ("Can the Shape of Nanoparticles Enable the Targeting to Cancer Cells over Healthy Cells?") they report successfully targeting breast and pancreas cancer cells over their healthy equivalents. | |
| Gooding explains that each cell type has its specific dominant uptake pathway(s) and each uptake pathway prefers a specific size or shape of nanoparticles to be internalized. "Comparing the uptake pathway of nanoparticles into cancer and healthy cells allow us to control the nanoparticles to get in the cancer cells and inhibit the nanoparticles to get into healthy cells." | |
| For example, they found that both breast and pancreas cancer cells as well as their healthy counterparts use the clathrin-mediated endocytosis pathway to take up nanorods. But only cancer cells use the macropinocytosis pathway to take up nanorods. | |
| By inhibiting the clathrin-mediated endocytosis pathway of both cancer and healthy cells, the researchers can make only the cancer cells take up nanorods via macropinocytosis pathway. | |
| To do this they employed hydroxychloroquine, a common inhibitor used to inhibit the clathrin-mediated endocytosis pathway. Plus, hydroxychloroquine is a classic drug and has been used for human for decades so their toxicity is well-documented. | |
 |
|
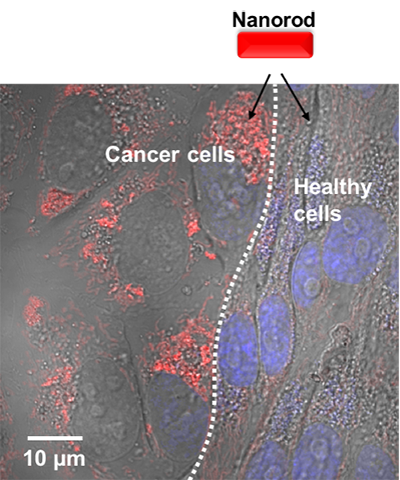
| A mixed co-culture of breast cancer and healthy cells (healthy cells are stained by cell tracker violet). The cells are treated with hydroxychloroquine before adding Cy.5.5-labelled nanorods (red emission). It can be seen that most nanorods are taken by cancer cells. (Image: Prof. Gooding, UNSW) | |
| "We think this is one of the first examples, if not the first, to use a combination of nanoparticle shape and uptake pathways used by cancer cells alone, to give selective targeting," Gooding points out. "Further, there are very few examples in the drug delivery field where the uptake of targeted drug delivery vehicles in cancer cells is compared with healthy cells. Cancer nanomedicine researchers heavily focus on cancer cells, but less so on healthy cells. We saw this as a big mistake, and also a big gap here to explore." | |
| The implication of this work is that features unique to cancers and the cancer microenvironment, other than surface features on cells, can be used to achieve selective targeting of nanoparticle drug delivery vehicles. This means that nanoparticle shape is an important factor to explore for targeting cancer cells and it is possible to design the shape of nanoparticles to selectively target cancer cells over healthy cells. | |
| Gooding and his team are now expanding their fundamental findings to practical applications. They are currently encapsulating anticancer drugs into mesoporous silica nanorods and use this nanostructure to deliver drugs to the cancer cells without harming the healthy cells (a manuscript of this work is in preparation). | |
| They are also looking into the industrial perspective. "Imagine if we can just adjust the shape of nanomedicine to make it enter the cancer cells only, it will significantly reduce the manufacturing cost and speed up the clinical translation," says Gooding. | |
| He notes that researchers in the nanomedicine field should rethink the active target strategy. "Active targeting using surface receptors is conferring as big an advantage as we had hoped in many cases. The reason for this may be related to an antibody modified nanoparticle contacting a healthy cell can still be taken up by the healthy cell. Instead, as we have shown here, a materials design approach to restrict nanoparticle uptake to endocytosis mechanisms only found in cancer cells can confer a considerable advantage." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
