| Feb 25, 2022 | |
Chemically programmed nanotranslators facilitate communication between microorganisms from different kingdoms |
|
| (Nanowerk Spotlight) Bacteria use chemical signals to communicate with one another through a process called quorum sensing, which allows a population of single-celled microbes to work like a multicellular organism. Originally discovered in the early 1970s in unusual bioluminescent marine bacteria, it is now believed that all bacteria 'talk' to one another with chemical signals. | |
| In a similar vein, organisms within other kingdoms use similar, well-defined signaling pathways to communicate with a member of the same species (i.e., pheromones in the animal kingdom or mating factors in fungi). However, these communication pathways are not shared with members from other kingdoms and organisms of different kingdoms do not usually communicate. | |
| "The design of chemical communication networks at the micro/nanoscale is an emergent interdisciplinary topic with potential applications in diverse areas such as sensing, biomedicine, biotechnology, and information and communication technologies," Ramón Martínez-Máñez tells Nanowerk. "Previously, researchers have demonstrated communication between synthetic microvesicles and one type of cells," adds Antoni Llopis-Lorente. "Communicating cells from different kingdoms has not been achieved before." | |
| Martínez-Máñez and Llopis-Lorente are co-authors of a recent paper in Nano Letters ("Nanoprogrammed Cross-Kingdom Communication Between Living Microorganisms") where a team of scientists from the Polytechnic University of Valencia report a proof-of-concept study that appears to be the first realization of a programmed cross-kingdom communication involving two species of living cells enabled by nanoparticles. | |
| In this work, the team employs tailor-made nanoparticles – mesoporous silica nanoparticles loaded with a molecular messenger (phleomycin) and capped with a glucose oxidase (GOx)-based responsive gatekeeper – to act as 'nanotranslators' to enable cross-kingdom communication between living microorganisms that otherwise would not interact. | |
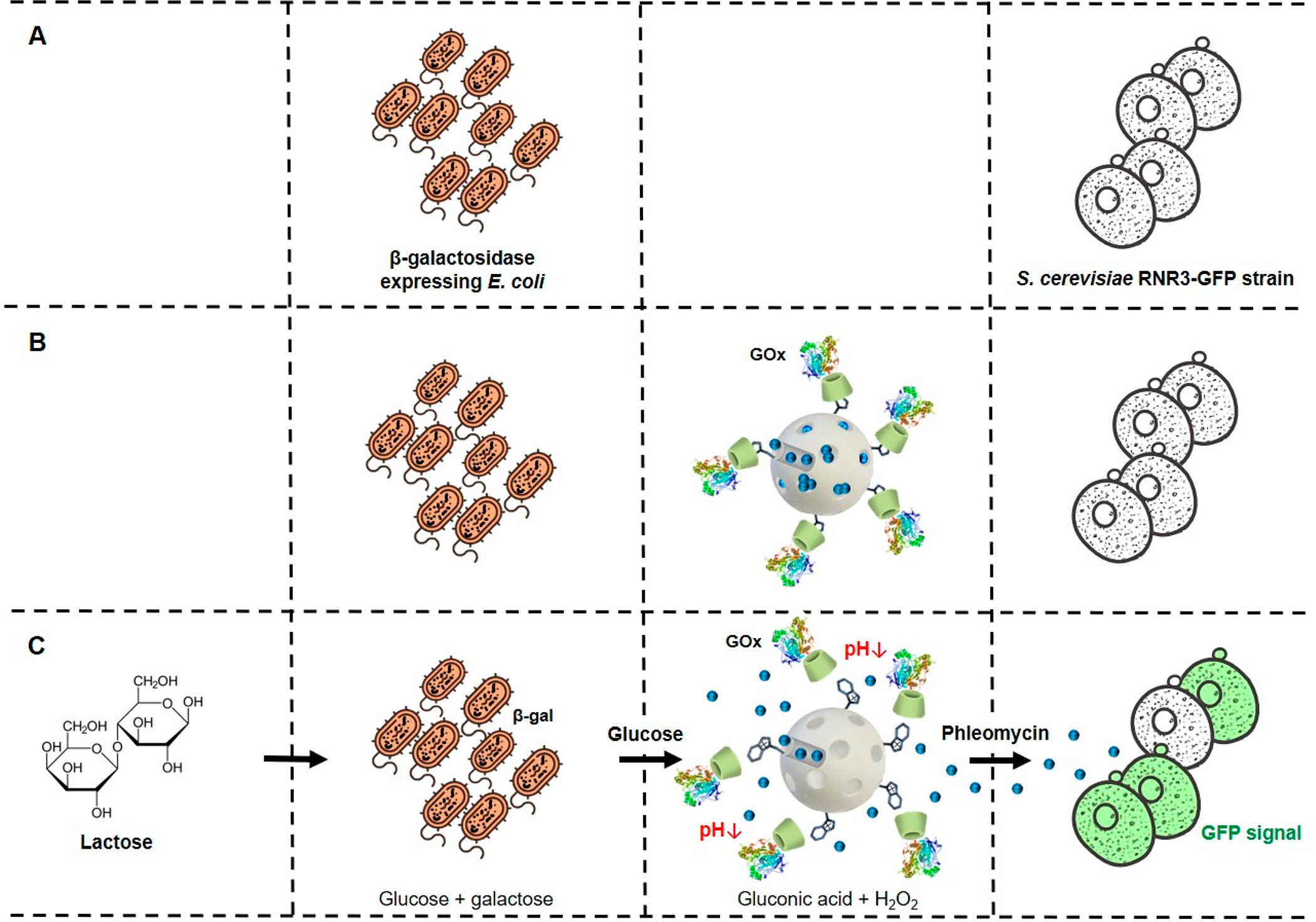 |
|
| (A) E. coli (β-galactosidase-expressing) bacterium cells do not communicate with S. cerevisiae yeast cells under normal conditions. (B) Tailor-made mesoporous nanoparticles (loaded with phleomycin and capped with a GOx-based responsive gatekeeper) are added to enable communication. (C) Communication steps: bacterium cells convert lactose into glucose and galactose; glucose (first chemical messenger) is detected by the nanodevice inducing delivery of the entrapped phleomycin (second chemical messenger); finally, the receiver yeast cells sense phleomycin and respond by activating expression of GFP. | |
| As illustrated above, the proof-of-concept engineered scheme works like this: Molecular information from the environment (lactose) is processed by β-galactosidase-expressing E. coli bacteria (prokaryotic cells, bacteria kingdom) and transformed into a chemical signal (glucose). The glucose is detected by the nanoparticles, which translate the chemical message 'glucose' to the chemical messenger 'phleomycin', which can be understood by the receiver microorganism (S. cerevisiae – eukaryotic cells, fungi kingdom). In response to phleomycin, S. cerevisiae yeast cells activate a genetic cascade that leads to green fluorescent protein expression as the output of the communication. | |
| "The key is the engineering of nanoparticles with a receiver-sender role," Llopis-Lorente points out. "We have achieved this by identifying the key messengers in the communication of the respective cells and by designing responsive porous nanoparticles loaded with a cargo and equipped with enzymatic units. The enzyme units act as processor of the information and upon recognition of the first chemical message, induce the opening of the nanoparticle pores and the release of an entrapped chemical, i.e. the second chemical messenger)." | |
| The researchers envision a wide range of applications based on the use of engineered nanotranslators to enable communication between different types of cells or microorganisms. For instance, designing nanoparticles that can enable plants and fungi talk to each other could facilitate the development of new ways to protect plants, new agricultural methods and eco-friendly ways to protect the environment. | |
| A quite intriguing prospect for medical and health applications is the possibility of regulating the interactions between bacteria and human cells, for instance to prevent infections, kill harmful bacteria, modulate the intestinal microbiome, or regulate interactions between cancer cells and immune cells. | |
| The team says that future directions of this work are the design of tailor-made nanoparticles to enable different types of communication, the demonstration of more complex communication networks, and the validation of communication in complex settings such as in vivo. | |
| "Perhaps the biggest challenge will be to read out if the communication has been successful or not," Martínez-Máñez concludes. "In the present work, we have used the expression of green fluorescent protein by the receiver cells, which facilitated visual monitoring the process. The development of future applications will require more advanced methodologies to enable the monitoring of chemical communication processes in complex biological settings such as in vivo or in agricultural settings. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
