| Mar 29, 2023 | |
Accelerating diabetic wound healing with innovative metal selenide ROS biocatalysts |
|
| (Nanowerk Spotlight) Diabetic ulcers, a serious complication of diabetes, can substantially increase amputation rates and mortality for diabetic patients. The inefficient healing of these ulcers is primarily due to the presence of high levels of reactive oxygen species (ROS) in the wound, contributing to oxidative stress and inflammation. This environment negatively affects the proliferation and differentiation of stem cells, hindering the healing process. | |
| Researchers have been working on developing materials to eliminate ROS and alleviate oxidative stress, including antioxidant nanostructures and ROS-scavenging materials that mimic the functions of natural antioxidases, such as catalase (CAT), glutathione peroxidase (GPx), and superoxide dismutase (SOD). | |
| However, as Prof. Chong Cheng, a full professor and a nanomaterial specialist at College of Polymer Science and Engineering, State Key Laboratory of Polymer Materials Engineering in Sichuan University, explains to Nanowerk, current synthetic metal compound-based biocatalysts have difficulty mimicking the complex biochemical process of natural antioxidases. This results in low catalytic efficiency, necessitating high dosages. | |
| To overcome this challenge, Prof. Cheng’s team recently constructed a cobalt selenide-based biocatalyst with an amorphous Ru@CoSe nanolayer for ultrafast and broad-spectrum catalytic ROS-elimination. | |
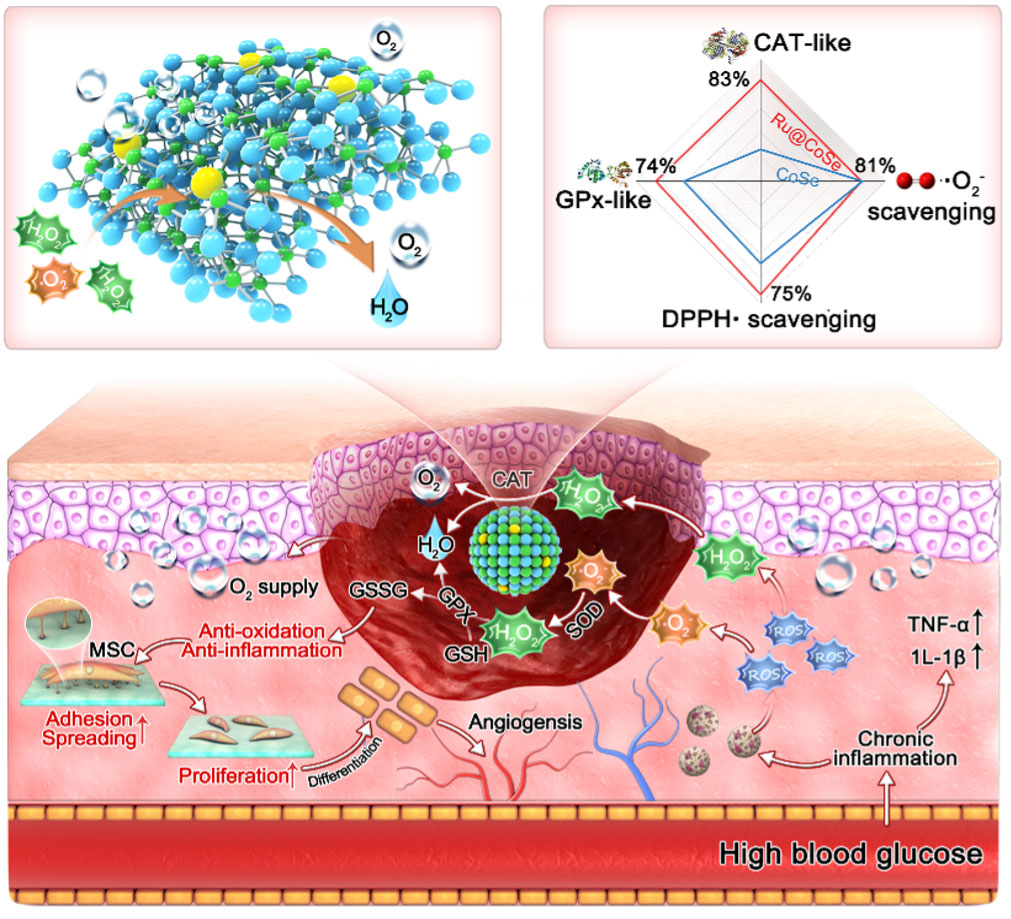 |
|
| Figure 1. Illustration of the working mechanisms of Ru@CoSe in scavenging ROS, protecting stem cells, and promoting angiogenesis. (Image courtesy of the researchers) | |
| This novel approach, published in ACS Nano ("Amorphizing Metal Selenides-Based ROS Biocatalysts at Surface Nanolayer toward Ultrafast Inflammatory Diabetic Wound Healing"), regulates the electronic structure and reversible redox properties of metal selenides for the first time. By incorporating enriched electrons and more unoccupied orbitals of Ru atoms into the CoSe matrix, the researchers achieved ultrafast and efficient ROS-scavenging activity. | |
| The Ru@CoSe biocatalyst exhibits superior CAT-like reaction kinetics (maximal reaction velocity: 23.05 µM s-1, turnover number: 2.00 s-1) and ultrafast, efficient ROS-scavenging activity. | |
| In both in vitro and in vivo experiments, the Ru@CoSe biocatalyst significantly reduces intracellular ROS concentration, alleviates hypoxia, and enhances human mesenchymal stem cell (hMSC) proliferation and angiogenic potential. These results lead to accelerated tissue regeneration and healing of inflammatory diabetic ulcers. | |
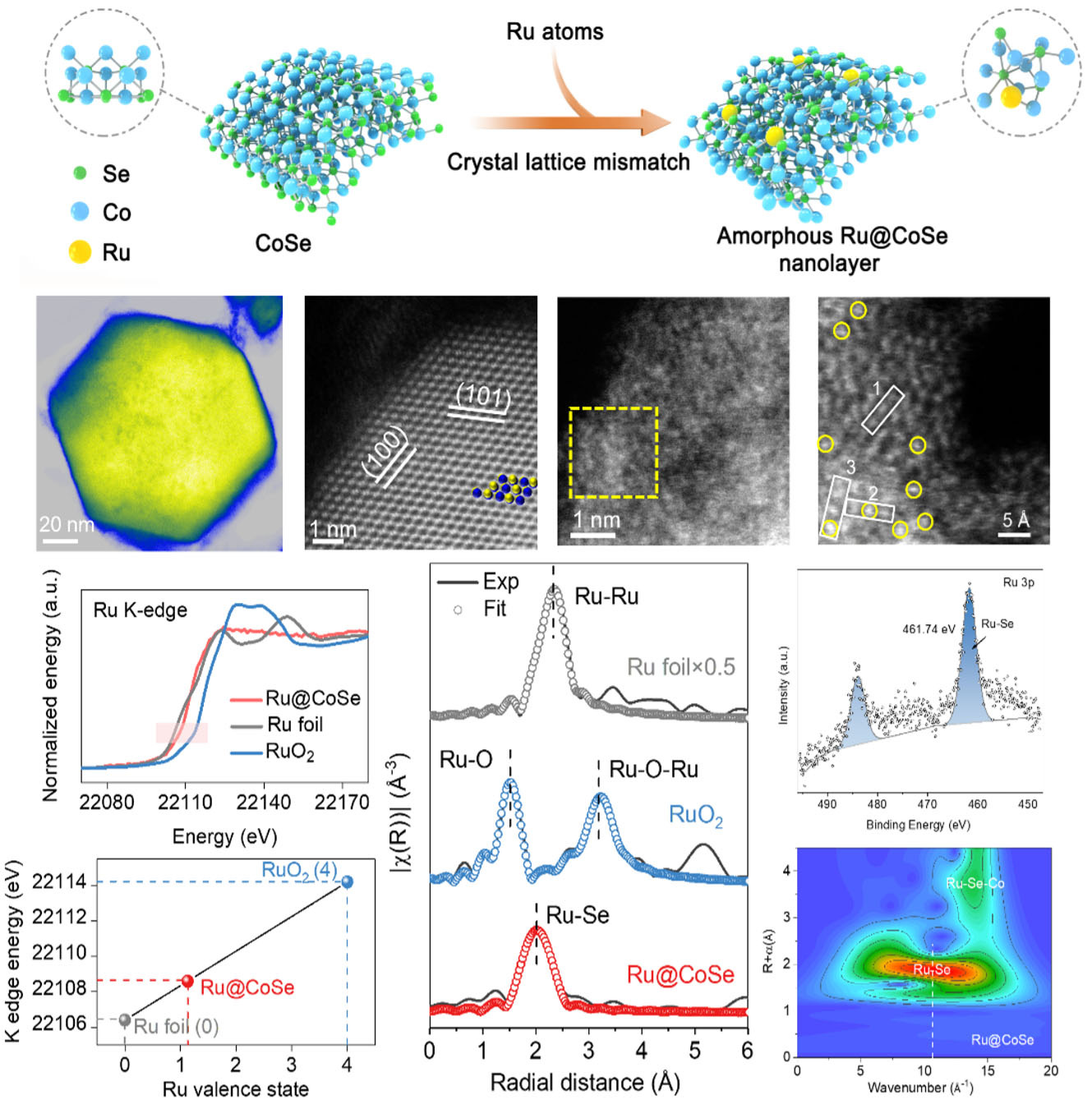 |
|
| Figure 2. The structural characterization of Ru@CoSe biocatalyst with amorphous nanolayer. (Image courtesy of the researchers) | |
|
Prof. Cheng emphasizes that their study presents an effective nanomedicine for catalytic ROS-scavenging and ultrafast healing of inflammatory wounds.
"Additionally, our research offers a strategy for designing biocatalytic metal compounds using amorphous catalytic structures," he concludes. "This groundbreaking work holds the potential to pave the way for developing artificial biocatalysts to treat chronic inflammatory diseases, ultimately improving the lives of millions of diabetic patients worldwide."
|
|
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
