| Nov 20, 2023 | |
Bioengineered viruses enable quantum leaps in biopyroelectric sensing and energy harvesting |
|
| (Nanowerk Spotlight) For decades, researchers have sought to understand and harness the pyroelectric effect in biological materials. Pyroelectricity refers to the phenomenon where heating or cooling a material with a nonzero spontaneous electric polarization changes the polarization, generating a temporary voltage. Many biological substances like bone, tendon, skin, and even cells exhibit weak pyroelectric properties. | |
| However, the inherent complexity of natural biomaterials has stymied attempts to elucidate the molecular mechanisms behind their pyroelectric behaviors. Manipulating biological structures at the nanoscale to optimize their pyroelectric performance has proven even more challenging. | |
| Recent advances in synthetic biology and genetic engineering now offer tantalizing possibilities for custom-designing biological structures with enhanced pyroelectric attributes. Viruses provide an especially promising platform. Their replicative nature allows mass-producing uniform pyroelectric materials encoded by engineered genes. | |
| Viruses also permit exquisite control over dipole alignment and polarization, which strongly influence pyroelectric output. However, while prior studies demonstrated piezoelectricity and triboelectricity in genetically altered viruses, none have successfully induced or measured pyroelectricity until now. | |
| In a new study (Advanced Materials, "Virus-Based Pyroelectricity"), researchers from UC Berkeley and Lawrence Berkeley National Laboratory report the first virus with sizable, tunable pyroelectric effects. The team chose the M13 bacteriophage as their testbed virus. M13 possesses a filamentous shape and helical protein coat that lacks inversion symmetry, conferring an intrinsic polarization. By inserting charged amino acids into its major coat proteins through gene editing, the researchers could adjust the phage’s polarization strength. Binding engineered minor proteins to nickel-coated substrates enabled orienting the phages vertically in dense aligned films with their polar axes uniformly pointing up. | |
 |
|
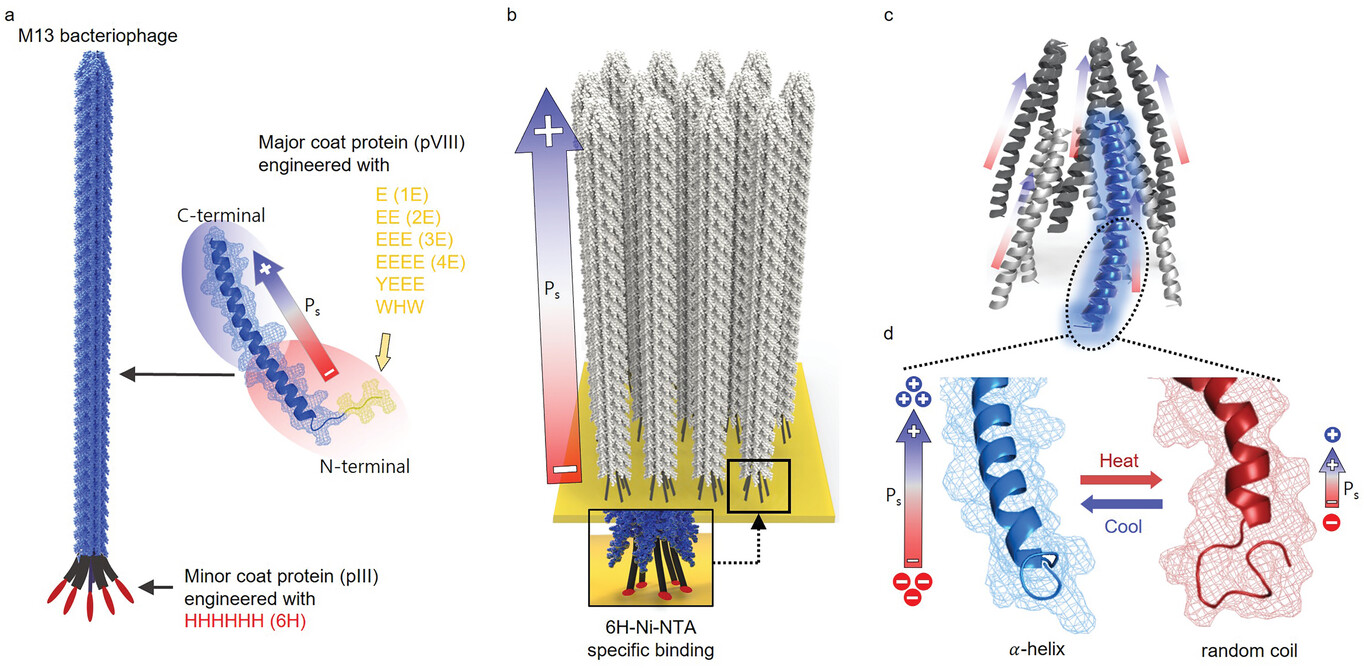
| Schematic diagram of M13 phage and its pyroelectric characteristics. a) M13 phage is a filament virus measuring 880 nm in length and 6.6 nm in diameter. It is composed of 2700 copies of pVIII major coat proteins and five copies of pIII minor coat proteins. b) Schematic representation of a vertically standing M13 phage film. The phage film exhibits a unidirectional arrangement of spontaneous polarization (Ps) achieved by introducing specific binding between the pIII proteins and the substrate using 6H-Ni-NTA. c) Schematic depiction of one repeating unit (consisting of 10 pVIII proteins) of the M13 major coat proteins. The pVIII proteins form a non-centrosymmetric structure characterized by a five-fold rotational and two-fold screw symmetrical arrangement. d) The pVIII protein possesses spontaneous polarization attributed to its α-helical protein structures. Upon transformation of the secondary structure into a random coil structure, the spontaneous polarization decreases. (Reprinted with permission by Wiley-VCH Verlag) | |
| Heating the vertically aligned phage films generated measureable pyroelectric potentials, detected via Kelvin probe force microscopy. The magnitude of the voltage correlated with the degree of polarity enhancement by genetic insertions. Of the modified phages, those engineered with four extra negative charges on each major coat protein gave the strongest pyroelectric coefficient – 0.13 µC m−2 °C−1. This substantially exceeds the 0.01-0.05 µC m−2 °C−1 range typical of natural biological tissues. | |
| Molecular dynamics simulations revealed that increasing temperatures unfolded the alpha-helical sections of the major coat proteins, diminishing their dipole moments. This molecular mechanism of pyroelectricity was confirmed by circular dichroism spectroscopy, which tracked the loss of alpha-helical structures when heating the phages. | |
| Besides proving viruses can leverage protein conformational changes to give robust pyroelectric outputs, the results provide much-needed insights into the microscale origins of biopyroelectricity. | |
| The researchers also showed the engineered phages’ pyroelectric currents could sensitively detect volatile organic compounds through peptide motifs on the viral coat specially designed to bind target molecules. Phages displaying hydrophilic peptides generated currents proportional to the surrounding humidity and polarity of exposed organic solvents. Meanwhile, phages exhibiting peptides that interact with aromatic compounds selectively produced currents when detecting benzene, toluene and xylene fumes. | |
| This pioneering demonstration of substantial, tunable pyroelectricity in a genetically engineered virus opens doors for numerous applications. By replicating virus-based pyroelectric materials en masse, lightweight biodegradable pyroelectric energy harvesters could be affordably manufactured. | |
| Implanted biopyroelectric devices may one day draw electrical power from natural heat gradients inside the human body. Virus-based pyroelectric gas and vapor sensors also offer inexpensive yet highly sensitive alternatives to existing electronic devices. | |
| More broadly, this research illuminates new prospects for engineering superior pyroelectric biomaterials. Insights gained from modifying virus structures could guide enhancements of pyroelectric properties in tissues, cells and proteins. Rather than relying on natural biomolecules, purpose-built biopyroelectric materials synthesized through synthetic biology may fulfill needs from compact infra-red sensors to flexible biomedical monitors benefiting human health and industry. | |
| Just as optogenetics revolutionized neuroscience by imparting light-sensing abilities to neurons, imparting pyroelectric capabilities to biologicals via genetic engineering could spur countless inventions benefiting society. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
