| Dec 15, 2023 | |
New bioink allows bioprinting directly onto wet, irregular surfaces |
|
| (Nanowerk Spotlight) Biofabrication holds incredible promise for regenerating damaged tissues and organs. Yet a fundamental challenge has hindered progress: conventional bioinks designed to print living cells cannot cling to moist, irregular wound surfaces. Without immediate, seamless adhesion, intricate bioprinted structures collapse upon contact with tissue in the very sites they aim to repair. | |
| Now, researchers have formulated an adhesive bioink to overcome this barrier. Composed of gelatin, frog skin secretions, and other polymers, the new ink displays unmatched wetting properties. As detailed in a recent Small paper ("An Adhesive Bioink toward Biofabrication under Wet Conditions"), it bonds firmly to tissues and diverse surfaces when printed underwater—retaining precision shape fidelity. | |
| This wet-adhesive bioink signals a major advance toward stabilizing delicate bioprinted tissue grafts within dynamic, moist wound environments. The technology introduces new possibilities for bioprinting directly onto or into organs during surgical procedures. It also opens the door to printing living bandages or skin patches. | |
 |
|
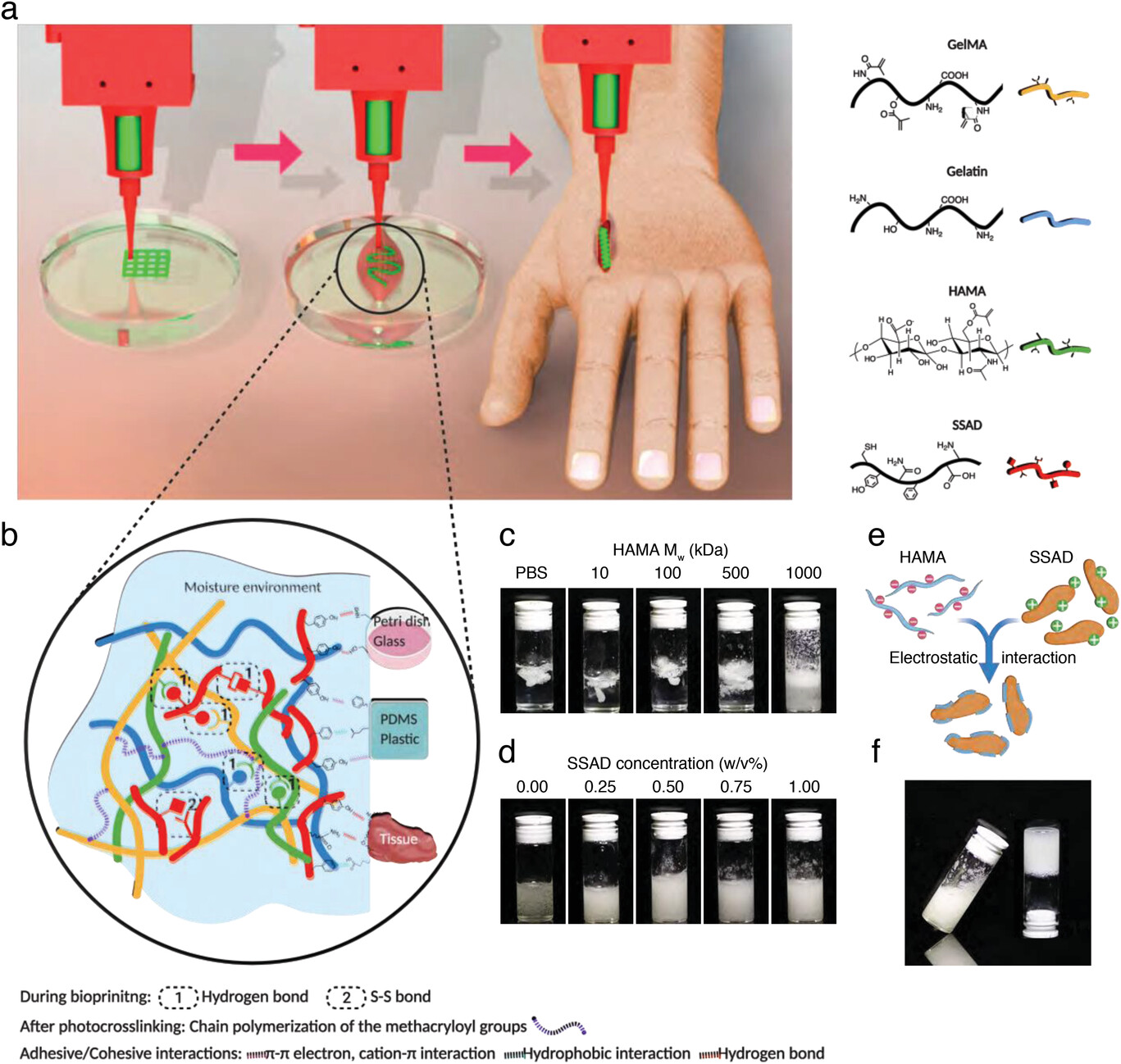
| Illustrations of extrusion bioprinting under wet conditions and the composition of the adhesive/cohesive bioink. a) A diagram of the bioprinting process, including bioprinting in wet conditions, testing in vitro, and in situ bioprinting. b) The interactions within the bioink include primarily S–S bond and hydrogen bonds, which contributes to the bioink cohesiveness. The chain polymerization of GelMA and HAMA after photocrosslinking further introduces stable chemical bonds. The adhesive mechanisms include π-π electron, cation–π interaction, hydrophobic interactions, hydrogen bonds, as well as metal coordination bonds between the deposited bioink and different substrates, including tissue culture-treated petri dish, glass, PDMS, plastic (pristine polystyrene), and biological tissues. c) SSAD distribution in HAMA solutions with different Mw under the same concentration (1.0 w/v%). d) SSAD distribution in HAMA (Mw = 1000 kDa) solutions with different SSAD concentrations. e) Schematic showing negatively charged HAMA neutralizing positively charged SSAD through electrostatic interactions. f) Photograph of the selected bioink with well-distributed SSAD. (Reprinted with permission by Wiley-VCH Verlag) | |
| The study details an adhesive bioink containing gelatin methacryloyl (GelMA), gelatin, methacrylated hyaluronic acid (HAMA), and a natural bioadhesive polymer from frog skin secretions. While previous bioinks lacked strong interlayer and surface bonding in wet environments like surgical settings, this new ink displays versatile adhesion and high shape fidelity when printed on diverse tissues, implants, and other surfaces. | |
| Recreating complex living structures requires printing multiple materials and cell types into intricate 3D architectures. However, standard bioinks often disintegrate when printed underwater. This presents obstacles for emerging techniques like intraoperative bioprinting, where tissue grafts are directly printed into wound sites during surgery. The research team set out to formulate a wet-adhesive ink to address this problem. | |
| They found that integrating frog skin secretions with GelMA, gelatin, and HAMA produced an ink with enhanced cohesive and adhesive properties. GelMA forms the photocrosslinkable scaffold, while gelatin Provides space for cell migration. HAMA helps disperse the frog skin particles for optimal adhesive effects. | |
| Through electrostatic and hydrophobic interactions, HAMA’s negative charge stabilizes the positive charge of the bioadhesive frog skin secretions. This prevents particle clumping and evenly distributes adhesiveness. Together, hydrogen bonding among all components enables excellent ink flow and shape retention when extruded underwater. | |
| The ink displays shear-thinning viscosity ideal for bioprinting, rapidly solidifying after extrusion. Lap shear adhesion strength reaches up to 17 kPa on porcine skin with wet surfaces—over twice as high as control bioinks. Importantly, strong adhesion occurs on many tissues and materials like glass, plastic, and silicone under wet conditions. | |
| Using a customized bioprinting system, the researchers showcased multi-interface printing of sophisticated 2D and 3D architectures underwater. Pattern fidelity remains unchanged in various media like cell culture medium, demonstrating versatility. To enhance precision, maneuverability and access for surgery, they integrated the bioink with handheld bioprinting tools. | |
| Complex flat and 3D constructs were successfully printed in situ onto ex vivo porcine skin using different ergonomic nozzles. Encapsulated NIH/3T3 fibroblasts exhibit over 95% viability immediately after wet printing, proliferating over 5 days of culture. In rats, subcutaneous hydrogel implants display sustained structural integrity and biocompatibility over 4 weeks. | |
| Finally, the bioink’s rapid gelation and tissue adhesion enabled effective hemostasis in liver wounds. Applied to bleeding incisions in rats, the ink quickly stopped blood loss compared to both commercial fibrin glue and untreated controls. | |
| With strong wet adhesion, high biocompatibility, and adaptable fluid properties, this new bioink formula shows immense promise. Handheld bioprinting may now enable intricate tissue grafts to be printed and adhere directly during surgery. The technology could also produce tissue patches, bandages, and hemostatic products. | |
| Ongoing challenges include vascularizing thicker printed tissues and preventing postoperative graft detachment due to movement. Integrating controlled drug release may help enhance cell survival and integration. Nonetheless, this adhesive ink should open new horizons for bioprinting in regenerative medicine and wound treatment. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
