| Dec 28, 2023 | |
MXenes, bare MXenes, or MX: The quest for the ideal catalyst |
|
| (Nanowerk Spotlight) Inexpensive and Earth-abundant alternatives to expensive noble metal catalysts remain a grand challenge at the forefront of the chemical industry. Catalysts play an indispensable role in the vast majority of industrial chemical processes by accelerating reaction rates and enabling important transformations under feasible conditions. | |
| However, the gold-standard catalysts - platinum and other rare platinum group metals - continue to constrain widespread adoption and scale-up in key sectors like fuels, bulk chemicals and pharmaceuticals due to supply limitations and exorbitant cost. This persisting need has recently fueled significant interest in a promising new class of materials called MXenes. | |
| This article explores the insights and findings from a perspective paper (Advanced Materials, "MAX, MXene, or MX: What Are They and Which One Is Better?") by researchers at National University of Singapore, focusing on the advancements in MXenes and their potential impact on catalysis and energy sectors. | |
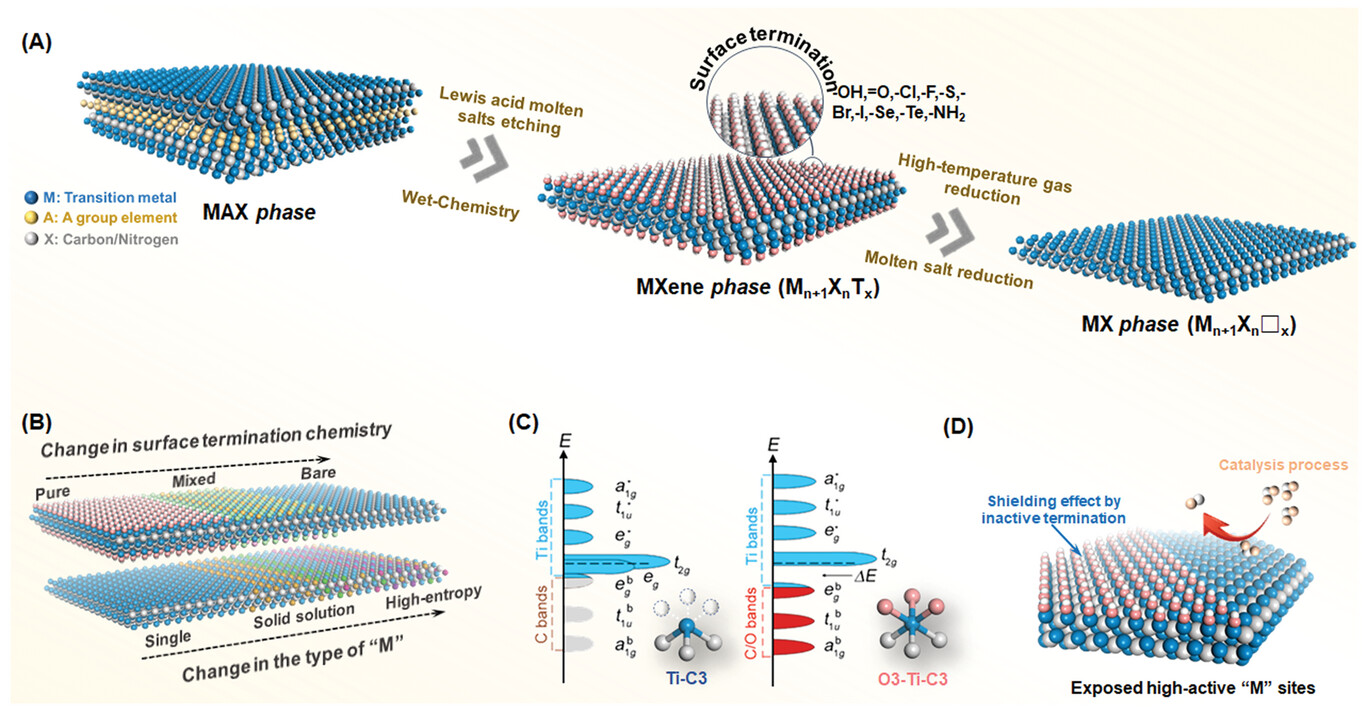 |
|
| A) Schematic illustration of structure from MAX to MXene, and then MX; B) Schematic illustration of current research tendencies in MXenes from different compositional and structural factors determining intrinsic properties of MXenes; C) Local atomic coordination around surface Ti and schematic of the orbitals for Ti−C3 in bare Ti3C2 and Ti−C3O3 octahedra in Ti3C2O2. The black dashed line at 0 eV represents EF; D) Schematic illustration of the comparison of the surface active site in terminated MXene and bare MX. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
| The journey of MXenes began with the exploration of their potential as catalysts, a critical component in numerous industrial processes. The global chemicals market, valued at over US $5 trillion in 2023, heavily relies on catalytic processes, with an estimated 85% of chemical products involving catalysis. | |
| MXenes, however, emerged as a promising alternative, offering similar catalytic properties to noble metals. This class of materials, derived from the etching of aluminum layers from MAX phases (where 'M' stands for a transition metal, 'A' for an element like aluminum, and 'X' for carbon and/or nitrogen), presents an intriguing and tunable electronic structure, rich surface-active sites, and high thermal durability. These properties make MXenes particularly attractive for a variety of applications, including energy storage and conversion, sensors, and, most notably, catalysis. | |
| However, an Achilles’ heel has hampered MXenes’ catalysis applications: surface terminations. MXenes form spontaneously when etching layered MAX phase precursors in aqueous fluoride solutions. The etching process leaves residual functional groups like –OH, –F and –O terminated on MXenes’ surfaces. Researchers have demonstrated that these terminations significantly degrade MXenes’ catalytic activities by blocking active sites, altering electronic band structures and raising reaction barriers. | |
The Leap Forward: From MAX to MXene to MX |
|
| The evolution from MAX phases to MXenes, and further to termination-free MXenes (referred to as MX), is a key focus of current research. | |
| Removing surface terminations to create “bare” or termination-free MXene (MX) drastically enhances predicted catalytic performance. Bare MX surfaces teem with fully exposed, unsaturated active metal sites ready to interact with adsorbates. MX also exhibits metallic conductivity and thermal stability up to 500 °C, ideal properties for high-temperature gas-phase catalysis. | |
| First-principles simulations show MX strongly chemisorbs intermediates and transition states in reactions including CO2 hydrogenation, water-gas shift and ammonia synthesis. An early 2021 study experimentally confirmed bare Mo2C MXene’s superior CO2 hydrogenation kinetics over termination-covered Mo2CTx MXene. Nevertheless, lack of scalable synthesis protocols and concerns about MX’s aqueous stability have obstructed further catalysis research. | |
| The perspective paper makes the case that these roadblocks are surmountable. MXenes’ surface terminations desorb above 400 °C, suggesting high-temperature MX formation is possible. MX should also remain stable for gas-phase reactions like CO2 hydrogenation. The authors argue MX’s prospects in heterogeneous catalysis are bright, especially as research transitions from lab-scale syntheses to industry-amenable production. | |
| Unique advantages prime MX for other applications beyond catalysis. In batteries, bare MXene strongly interacts with polysulfides compared to functionalized MXene, promising improved lithium-sulfur performance. Lewis acidic metal sites may also allow MX to electrochemically reduce dinitrogen to ammonia mieux mieux than surface-blocked MXene electrocatalysts evaluated thus far. | |
| Further tailoring through compositional modifications like high-entropy and single atom alloying could elicit superior catalytic properties. Multiple metals’ synergistic effects and lattice strain in high-entropy MX may lower reaction barriers and binding energies. Anchoring individual platinum, ruthenium or iridium atoms onto MXenes could likewise yield highly uniform, maximized distributions of precious metal active sites. Realizing these concepts will require resolving nagging MX synthesis obstacles first. | |
| By collating emerging theoretical and experimental evidence, the review builds a compelling case for bare MXenes’ untapped potential in next-generation thermal catalysis. If key bottlenecks around production scaling, surface stabilization and real-world performance can be addressed, MXenes may finally fulfill their promise as widely available, high-performance catalyst alternatives. Though the path ahead remains challenging, rapid progress in MXene research over the past decade inspires optimism that these ultra-thin materials could catalyze transformative breakthroughs across the chemical industry. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
