| Posted: Aug 20, 2008 | |
Sunburn increases risk of nanoparticle skin penetration |
|
| (Nanowerk Spotlight) A couple of weeks ago we posted a commentary by Samuel S. Epstein, Professor Emeritus of Environmental and Occupational Medicine at the University of Illinois at Chicago School of Public Health and Chairman of the Cancer Prevention Coalition, who argued that the U.S. Food and Drug Administration (FDA) has failed to properly regulate potentially dangerous sunscreens (FDA remains asleep at the wheel on dangers of sunscreens, besides other cosmetics and personal care products). | |
| In similar fashion, consumer and environmental advocacy groups like Friends of the Earth (who published a report in May 2006 that takes a very critical look at nanomaterials in cosmetics: "Nanomaterials, Sunscreens and Cosmetics; Small Ingredients, Big Risks"") are calling for application of the Precautionary Principle to manufactured nanoparticles and other applications of nanotechnology. Here, the basic idea is that manufacturers should have to prove to regulators that their products are not harmful before the products are sold (see our Spotlight: "Late lessons from early warnings for nanotechnology "). | |
| The way things stand now, nanotechnology products can be sold unlabeled and the FDA regulates sunscreens only based on their sun protection factor (SPF). Cosmetic manufacturers, of course, claim that their products, including nanoparticle-based sunscreens are harmless. Indeed, nobody has demonstrated that they are unsafe – but the opposite proof, that they are perfectly safe, is missing as well. This confusing situation is due to the incomplete scientific picture created by a lack of relevant research. For instance, the question of whether or not nanoparticles can penetrate the healthy stratum corneum skin barrier in vivo remains largely unanswered. Furthermore, no studies so far have examined the impact of ultraviolet (UV) radiation on nanoparticle skin penetration. Since sunscreen is often applied to sun damaged skin, such a real world scenario, as opposed to in vitro studies in a test-tube, could go a long way in confirming or allaying fears. | |
| New research by scientists at the University of Rochester is the first to consider the effects of nanoparticle penetration through normal and barrier defective skin using an in vivo model system. | |
| "Our work was motivated by the increasing occurrences of occupational and consumer nanoparticle skin exposures and the fact that in vitro studies suggest that nanoparticles including quantum dots can exhibit dose and time dependent cytotoxic effects" Dr. Lisa DeLouise explains to Nanowerk. "In conducting this work we also hope to shed light on inconsistencies that exist in pre-existing literature with regard to nanoparticle skin penetration using ex vivo skin models." | |
| DeLouise, an Assistant Professor in the Department of Dermatology and Biomedical Engineering at the University of Rochester's (UR) Medical Center, together with her PhD candidate and the paper's first author Luke Mortensen and colleagues Prof. Günter Oberdörster and Prof. Alice Pentland demonstrate the importance of skin condition to effect the penetration of quantum dot nanoparticles in an in vivo mouse model. They have published their findings in the August 8, 2008 online edition of Nano Letters ("In Vivo Skin Penetration of Quantum Dot Nanoparticles in the Murine Model: The Effect of UVR"). | |
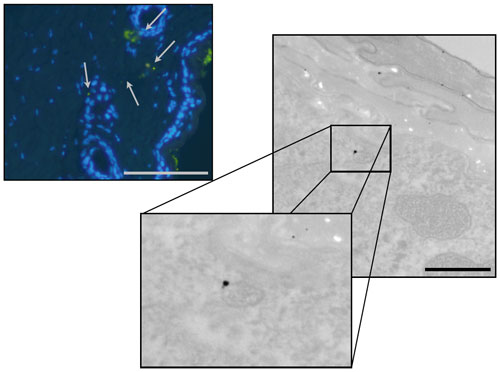 |
|
| Fluorescence and Transmission Electron Microscopy (TEM) images of quantum dot exposed skin 24 hours after application and UVR irradiation. Arrows point to instances of quantum dot penetration in the dermis that is believed to be localized near follicular openings. Fluorescence scale bar=50µm, TEM scale bar=1µm. (Images: Luke Mortensen, University of Rochester) | |
| The scientific core of the UR scientists' findings is that commercially available carboxyl-terminated quantum dot nanoparticles penetrate more readily through UV-damaged mouse skin than through undamaged skin. | |
| "Understanding the mechanisms and extent to which nanomaterials can penetrate skin is essential for establishing exposure health and safety guidelines" says DeLouise. "Our work advances this field as it documents that the outside-in barrier defect following an acute UV exposure permits quantum dot penetration. We found that the accelerated epidermal proliferation and differentiation of UV-radiated skin repair response is insufficient to prevent thequantum dots from breeching the skin barrier." | |
| The researchers point out, though, that their results show that only a very minute amount of the applied quantum dots penetrate UV-compromised skin. It is conceivalble that a small number of penetrated nanoparticles could cause adverse effects depending on their composition, nanoparticle in vivo solubility and routes of excretion. For the case of titanium dioxide and zinc oxide used in sunscreens, adverse side effects have not yet been documented, says DeLouise. This is in contrast to existing literature results of high-dose in vitro studies on skin cells that observe cytotoxic reactive oxygen species (ROS) generation. | |
| The researchers used quantum dots for their study because they enabled direct fluorescent imaging in skin. Quantum dots' efficient UV adsorbing and fluorescent properties has rapidly expanded their use in various biotechnology, military and security applications and they are being considered for use in cosmetic and UV blocking applications. Hence, it is imperative that these materials be designed to prevent skin penetration to avoid potential adverse health risk long term. | |
| The goal of the UR scientists is to fully characterize the effect of nanoparticle surface chemistry and size on the mechanisms and extent of penetration. | |
| "This knowledge will be used to rationally design nanoparticles to prevent or increase their penetration levels" explains DeLouise. "The ability to control nanoparticle penetration profiles will enable design of safe consumer products and will enable new biomedical technologies such as target nanoparticle therapeutic and diagnostic biomedical applications." | |
| To this end, the researchers are working with a new class of quantum dots based on lead selenide in collaboration with the Krauss Group at UR. Recent results of this work have been published in Nano Letters as well ("Ultrabright PbSe Magic-sized Clusters"). | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
