| Posted: Oct 08, 2008 | |
Live cell imaging with biodegradable quantum dot nanocomposites |
|
| (Nanowerk Spotlight) The study of individual live cells is a hugely important scientific task and essential to the field of molecular biology and biomedical research. Among the most significant technical challenges for performing successful live-cell imaging experiments is to maintain the cells in a healthy state and functioning normally on the microscope stage while being illuminated. Especially if scientists want to look into cellular processes that occur inside cells in their natural state and that cannot be observed by traditional cytological methods. | |
| Quantum dots (QDs), also called nanocrystals, hold increasing potential for in vitro and in vivo cellular imaging. For instance, we have previously reported about how researchers have used QDs for in vivo imaging of embryonic stem cells in mice, a novel technique that has opened up the possibility of using QDs for fast and accurate imaging applications in stem cell therapy (see: Quantum dot imaging could benefit embryonic stem cell therapy). | |
| The usefulness of quantum dots comes from their peak emission frequency's extreme sensitivity – quantum mechanical in nature – to both the dot's size and composition. QDs have been touted as possible replacements for organic dyes in the imaging of biological systems, due to their excellent fluorescent properties, good chemical stability, broad excitation ranges and high photobleaching thresholds. | |
| "Although QDs have been used quite successfully in imaging studies of both fixed and permeabilized cells, their use in live cell microscopy has been limited to staining and imaging of membrane-bound receptors and trafficking vesicles" Dr. Warren Chan explains to Nanowerk. "This drawback, which stems largely in part from difficulties in delivering large quantities of QDs into the cytoplasm without causing significant disruption to the plasma membrane integrity, has hindered the widespread use of QDs for tracking cytoplasmic proteins, organelles, and complex biomolecular processes in live cells." | |
| Chan, who is an Assistant Professor at the University of Toronto's Institute of Biomaterials and Biomedical Engineering, together with his group and researchers from UToronto's Terrence Donnelly Centre for Cellular and Biomolecular Research as well as the Division of Neurosurgery at UToronto's The Hospital for Sick Children, has developed a new strategy to deliver nanoparticles, in particular semiconductor quantum dots, into the cytoplasm (the contents of a cell except the nucleus) of a cell. | |
| By engineering a hybrid nanocomposite, the team has successfully delivered antibody conjugated QDs into the cytosol (the aqueous solution of a cell's cytoplasm) and demonstrated live cell labeling of cytoplasmic structures, including actin microfilaments, through the incorporation of anti-actin antibody conjugated quantum dots within the nanocomposite core. | |
| The scientists in Canada utilized a bio-degradable hydrogel system that carries the nanoparticles into the cytoplasm. The degradation of the hydrogel allows the nanoparticles to function, as engineered, inside the cell. For example, the nanoparticle could be used for labeling intracellular structures or delivery of drug to specific organelles – a task for which the nanoparticles must be delivered to the cytoplasm first. | |
| "Prior to our development, researchers attempted to use peptides for delivery but sometimes the peptides could be problematic for delivering bio-conjugated nanoparticles because the surface chemistry between the bio-conjugated molecule and peptide may not be amenable to cross the vesicle membrane to get into the cytoplasm" Chan remarks. "Other techniques include microinjection and electroporation but these techniques may not be high-throughput or could damage cells, respectively." | |
| Only one other technique, based on osmotic release of phagocytic vesicles, has shown high-throughput intracellular delivery. | |
| The novel technique than Chan and his collaborators demonstrate is a noninvasive method to image subcellular structures in live cells using bioconjugated QD nanocomposites. This delivery system is bioresponsive thanks to the incorporation of antibody-coated QDs within biodegradable polymeric nanospheres. Upon entering the cytosol, the polymer nanospheres undergo hydrolysis and thereby release the QD bioconjugates. | |
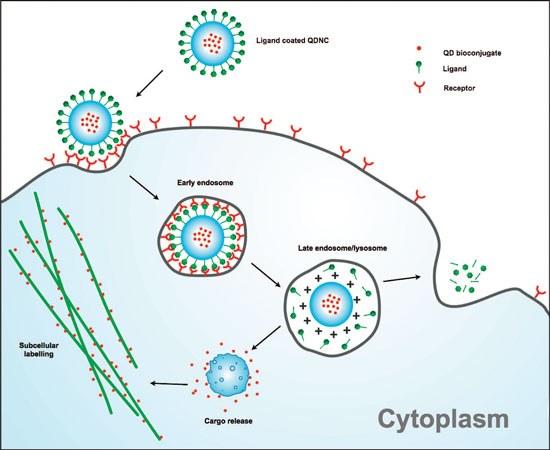 |
|
| Mechanism of cytosolic delivery and subcellular targeting of quantum dot nanocomposites (QDNCs). Schematic representation depicting QDNC escape from the endolysosomal compartment upon cellular internalization with cytosolic release of the encapsulated cargo. Antibody-conjugated QDs can be delivered in this manner to allow the labeling of subcellular organelles or other molecular targets. Note: The actual charge distribution on the QDNC may be heterogeneous inside the acidic vesicles with partial cleavage of the peptide bonds on the QDNC surface. (Reprinted with permission from American Chemical Society) | |
| Chan points out that utilizing the surface of biodegradable polymers for cell penetration, the chemical and structural properties of protein-coated QDs can be preserved, thus enabling them to actively seek specific intracellular targets within the cytosol. | |
| "The biocompatibility of the delivery system coupled with cell entry capabilities without disturbing the plasma membrane could extend the use of QD bioconjugates to selectively label subcellular structures, monitor and track cytoplasmic processes, and elucidate protein interactions in live cells" he says. | |
| According to Chan, the local targeting of specific subcellular organelles is necessary for the elucidation of fundamental process in live cells. He notes that the very same QD surface that is modified to enable intracellular delivery is also needed for the attachment of subcellular targeting biomolecules. | |
| "Achieving both tasks simultaneously by relying on surface chemistry of QDs alone will remain difficult" he says. "However, encapsulating QDs already coated with targeting molecules inside a capsule with a modifiable surface aimed at cytosolic delivery can circumvent such limitations. Once inside the cytosol, the capsule must release the QD bioconjugates in order to label subcellular structures through molecular recognition." | |
| Another potential for these nanocomposites is the ability to perform multiplexed staining through the simultaneous delivery and detection of multiple subcellular markers. Also, imaging of multiple cellular markers by encapsulating various wavelength-emitting quantum dots can be achieved. | |
| Chan notes that issues of cytotoxicity using cell penetrating strategies ultimately are dependent on the degree of membrane disruption; and both direct carrier injection and transient permeabilization of cell membrane using solution-based transfection agents have been reported to cause membrane damage. | |
| "We observed an enhanced biocompatibility of our nanocomposites as compared to free quantum dots" says Chan. "We think that the lack of significant disruption to membrane structures could help explain the minimal cytotoxicity of our QD composites." | |
| The team is now starting to conduct fundamental studies of the impact of the intracellular delivery of quantum dots on the cell system. "In order to truly use this technology, we have to really understand what it's doing to the cell" says Chan. | |
| This work was published in the September 25, 2008 online edition of Nano Letters ("Biodegradable Quantum Dot Nanocomposites Enable Live Cell Labeling and Imaging of Cytoplasmic Targets"). The first authors of the manuscript is Drs. Betty Kim and Wen Jiang and John Oreopoulos is a co-author. This project was in collaboration of Chan's group with the labs of Drs. James Rutka and Christopher Yip. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
