| Posted: Oct 25, 2006 | |
Understanding the workings of lipid-carbon nanotube self-assembly in water |
|
| (Nanowerk Spotlight) Back in 2005, Dr. Pu-Chun Ke conducted an experimental study ("Coating Single-Walled Carbon Nanotubes with Phospholipids") where he discovered a very efficient method of solubilizing nanotubes using lysophospholipids, or the so-called single-tailed phospholipids. | |
| The solubility provided by lysophospholipid LPC is at least one order of magnitude better than that provided by SDS, a routine surfactant people use to solubilize nanomaterials in aqueous solutions. Ke and his colleagues showed that superior solubility was due to the formation of lipid 'striations' coated on the nanotubes. The underling principle of this superior solubility of nanotubes by lysophospholipids is supramolecular assembly, a topic of common interest to researchers in nanoscience, chemistry, materials, and biophysics. | |
| New results obtained during recent follow-up research provide useful insight on the binding mechanism of amphiphiles and one-dimensional nanostructures. This knowledge may facilitate the bottom-up design of supramolecular assembly, nanotechnology, nanotoxicology, and gene and drug delivery. | |
| "Those images from the earlier study made us ponder about the nature of those striations" Ke, who is assistant professor at Clemson University's Department of Physics & Astronomy, explains to Nanowerk. | |
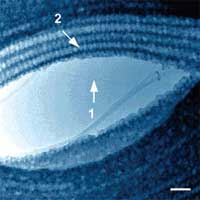 |
TEM image of SWNT-LPC complex. (1) an isolated SWNT in the vacuum phase. (2) an LPC striation on an SWNT/SWNT bundle. Scale bar 20 nm. (Reprinted with permission from American Chemical Society) |
| Supramolecular assemblies can be formed through weak intermolecular forces such as hydrophobic interactions, van der Waals, and electrostatic forces. Understanding supramolecular assembly such as lipid-nanotube binding has tremendous importance for the design of novel nanostructures and nanodevices for sensing, imaging, and nanomedicine. The other significance of understanding such a fundamental topic is for reliable and repeatable nanotoxicity studies (which are of paramount importance for the development of nanotechnology considering current nanotoxicity studies are phenomenological in general and are lack of guidance by fundamental research). | |
| There are three models people use to describe the binding of amphiphiles onto cylindrical nanostructures: hemimicelles, cylindrical surfactant encapsulation, and random adsorption. | |
| Schematic representations of the mechanisms by which surfactants help disperse SWNTs. (a) SWNT encapsulated in a cylindrical surfactant micelle: right: cross section; left: side view. (b) Hemimicellar adsorption of surfactant molecules on a SWNT. (c) Random adsorption of surfactant molecules on a SWNT. Source: "Small-Angle Neutron Scattering from Surfactant-Assisted Aqueous Dispersions of Carbon Nanotubes" (Reprinted with permission from American Chemical Society) | 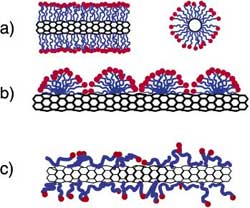 |
| "At the time of publishing the J. Phys. Chem. B paper we tended to think that the 'hemimicellar model' therein was the one to explain our TEM study but I was never comfortable with that idea because it was too 'mechanical' or too 'regular'" says Ke. | |
| In another set of experiments, the Clemson researchers utilized the method of solubilizing nanotubes with lysophospholipids ("Detection of phospholipid-carbon nanotube translocation using fluorescence energy transfer"). This time they attached a fluorophore rhodamine to lipid LPE ('Rd-LPE'). | |
| "The original idea was to use Rd-LPE to solubilize nantoubes and use Rd-LPE as a tracer to follow the motion of nanotubes in cells and in organisms for the purposes of drug and gene delivery" Ke explains. (The team first demonstrated the principle of RNA delivery using nanotubes in a 2004 paper in Nano Letters). "Once we mixed Rd-LPE with nanotubes we indeed obtained an excellent nanotube solubility but we found the fluorescence of the rhodamine was severely quenched. We interpreted that as a result of fluorescence energy transfer from the Rd dyes to the nanotubes (the latter, once solubilized, are known to absorb visible light and emit auto-fluorescence in near infrared)." | |
| Ke points out that they had accidentally created an optical switch which glows when Rd-LPE is dissociated from nanotubes and stops glowing once Rd-LPE is attached to the nanotubes. | |
| "In our cellular imaging study we found the Rd-LPE-nanotube assembly glowed only in cells but not outside the cells" notes Ke. "This tells us that the 'drug load' of Rd-LPE was released from the nanotubes in cells, possibly due to the disassembly by cell membranes in the process of their translocation. This 'on' and 'off' character of the Rd-LPE-nanotube complex can be used for drug delivery and biosensing." | |
| In the field of nanotoxicology this complex may be used to trace the multi-generational transfer and oxidative stress of nanomaterials. | |
| To understand the binding of lysophospholipids to nanotubes Ke collaborated with Dr. Rui Qiao, an assistant professor in Mechanical Engineering at Clemson. Using molecular dynamics simulations they found that none of the three models in the literature was accurate for describing the binding of an amphiphile molecule to a cylindrical nanostructure. The results are described in a recent article in the Journal of the American Chemical Society ("Lipid-Carbon Nanotube Self-Assembly in Aqueous Solution"). | |
| The two researchers found that the lipid tails prefer to lie down on the nanotube surface so that the energy cost for lipid bending would be minimal and the tolerance for other lipids in solution to bind onto the already adsorbed lipids would be maximum. They show in their paper that the periodicity of the 'striations' from their simulations is about 4.5 nm, matching very well with their TEM study. | |
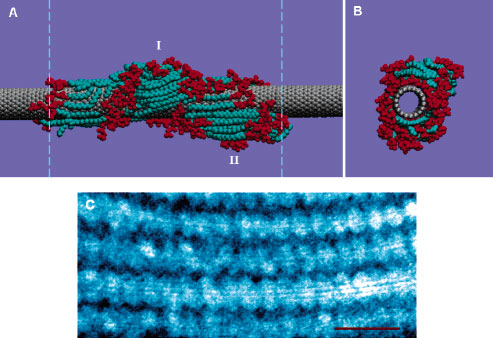 |
|
| Self-assembly of LPC and an SWNT. (A) Front view of the simulation system configuration at t=24 ns. The lipid bump I is formed from the gradual adsorption of lipids from the bulk, while the lipid bump II is formed from the adsorption of a lipid cluster in the bulk. The lipid headgroups and the tails are illustrated in red and cyan and the SWNT in gray, respectively. The dashed lines in panel A denote the periodic boundaries of the simulation box (∼12 nm). Panel B shows the side view of the SWNT-LPC simulation system (water not shown). (C) TEM image of SWNT-LPC assemblies display a striation periodicity of 4.5 nm: scale bar, 15 nm. (Reprinted with permission from American Chemical Society) | |
| Ke and Qiao now think that "obviously the cylindrical encapsulation model and the random adsorption model cannot explain the striations observed in the TEM experiments. The hemimicellar model requires lipid micelles first to break from the middle and then to assemble in tandem onto an SWNT. In comparison our illustrated mechanism agrees with the TEM experiments and is sterically and energetically favorable for the self-assembly of amphiphiles and cylindrical nanostructures." | |
| Ke believes that they have clarified an avidly debated subject on amphiphile-nanotube self assembly: "I think this study will provide an important guidance for researchers in the fields of materials synthesis and self assembly, alternative transporters for drug delivery, and novel nanodevices for biosensing and imaging. The future research in my lab in collaborations with my colleagues at Clemson and beyond will continue to explore, using experimental and in silico studies, the fundamental issues in integrating nanomaterials and biological systems. I believe our research is unique in that it examines nanomaterials in a biological setting from the angle of biophysics instead of chemistry or materials science, and I am hoping in the long run that our research will benefit the fields aforementioned including nanotoxicology." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
