| Posted: Jul 25, 2014 |
New study calls into question the existence of silicene
|
|
(Nanowerk News) Sometimes, scientific findings can shake the foundations of what was once held to be true, causing us to step back and re-examine our basic assumptions.
|
|
A recent study ("Silicon Growth at the Two-Dimensional Limit on Ag(111)") at the U.S. Department of Energy’s Argonne National Laboratory has called into question the existence of silicene, thought to be one of the world’s newest and hottest two-dimensional nanomaterials. The study may have great implications to a multi-billion dollar electronics industry that seeks to revolutionize technology at scales 80,000 times smaller than the human hair.
|
 |
| Argonne researchers use an e-beam evaporator to deposit atomic silicon onto a silver platform in a vacuum. The silver is heated to ~400 C allowing the deposits to rearrange into a sheet of interlocking silicon atoms. The B&W cross-sectional transmission electron microscope image to the right shows the growth of bulk-like silicon nanosheets, rather than atomically thin silicene layers.
|
|
Silicene was proposed as a two-dimensional sheet of silicon atoms that can be created experimentally by super-heating silicon and evaporating atoms onto a silver platform. Silver is the platform of choice because it will not affect the silicon via chemical bonding nor should alloying occur due to its low solubility. During the heating process, as the silicon atoms fall onto the platform, researchers believed that they were arranging themselves in certain ways to create a single sheet of interlocking atoms.
|
|
Silicon, on the other hand, exists in three dimensions and is one of the most common elements on Earth. A metal, semiconductor and insulator, purified silicon is extremely stable and has become essential to the integrated circuits and transistors that run most of our computers.
|
|
Both silicene and silicon should react immediately with oxygen, but they react slightly differently. In the case of silicon, oxygen breaks some of the silicon bonds of the first one or two atomic layers to form a layer of silicon-oxygen. This, surprisingly, acts a chemical barrier to prevent the decay of the lower layers.
|
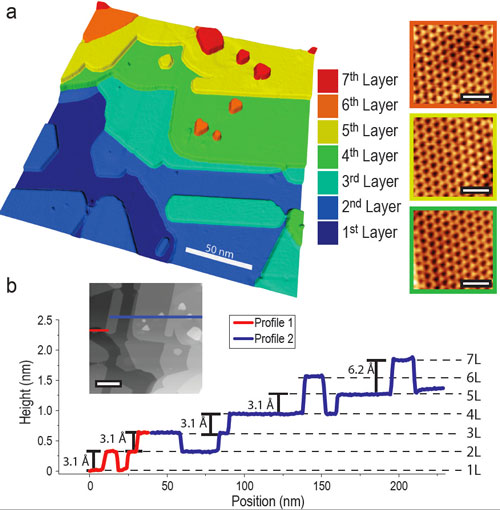 |
| The researchers find that silicene, a one-atom thick sheet of silicon, has not been experimentally realized on silver, and that silver is unlikely to be a good substrate for silicene. (a) This three-dimensional scanning tunneling microscopy image shows the growth of seven ultra-thin layers of silicon nanosheets atop a silver crystal. The atomic structures of the nanosheets are illustrated for layers 4, 5 and 6. (b) Step heights for silicon nanosheets are plotted at the bottom.
|
|
Because it consists of only one layer of silicon atoms, silicene must be handled in a vacuum. Exposure to any amount of oxygen would completely destroy the sample.
|
|
This difference is one of the keys to the researchers’ discovery. After depositing the atoms onto the silver platform, initial tests identified that alloy-like surface phases would form until bulk silicon layers, or “platelets” would precipitate out, which has been mistaken as two-dimensional silicene.
|
|
“Some of the bulk silicon platelets were more than one layer thick,” said Argonne scientist Nathan Guisinger of Argonne’s Center for Nanoscale Materials. “We determined that if we were dealing with multiple layers of silicon atoms, we could bring it out of our ultra-high vacuum chamber and bring it into air and do some other tests.”
|
|
“Everybody assumed the sample would immediately decay as soon as they pulled it out of the chamber,” added Northwestern University graduate student Brian Kiraly, one of the principal authors of the study. “We were the first to actually bring it out and perform major experiments outside of the vacuum.”
|
|
Each new series of experiments presented a new set of clues that this was, in fact, not silicene.
|
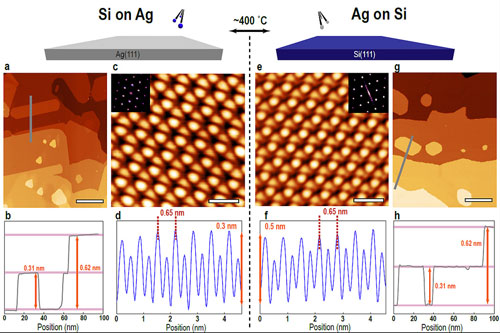 |
| Argonne researchers investigating the properties of silicene (a one-atom thick sheet of silicon atoms) compared scanning tunneling microscope images of atomic silicon growth on silver and atomic silver growth on silicon. The study finds that both growth processes exhibit identical heights and shapes (a, g), indistinguishable honeycomb structures (c, e) and atomic periodicity (d, f). This suggests the growth of bulk silicon on silver, with a silver-induced surface reconstruction, rather than silicene. (click on image to enlarge)
|
|
By examining and categorizing the top layers of the material, the researchers discovered silicon oxide, a sign of oxidation in the top layers. They were also surprised to find that particles from the silver platform alloyed with the silicon at significant depths.
|
|
“We found out that what previous researchers identified as silicene is really just a combination of the silicon and the silver,” said Northwestern graduate student Andrew Mannix.
|
|
For their final test, the researchers decided to probe the atomic signature of the material.
|
|
Materials are made up of systems of atoms that bond and vibrate in unique ways. Raman spectroscopy allows researchers to measure these bonds and vibrations. Housed within the Center for Nanoscale Materials, a DOE Office of Science User Facility, the spectroscope allows researchers to use light to “shift” the position of one atom in a crystal lattice, which in turn causes a shift in the position of its neighbors. Scientists define a material by measuring how strong or weak these bonds are in relation to the frequency at which the atoms vibrate.
|
|
The researchers noticed something oddly familiar when looking at the vibrational signatures and frequencies of their sample. Their sample did not exhibit characteristic vibrations of silicene, but it did match those of silicon.
|
|
“Having this many research groups and papers potentially be wrong does not happen often,” says Guisinger. “I hope our research helps guide future studies and convincingly demonstrates that silver is not a good platform if you are trying to grow silicene.”
|



