| Posted: Oct 18, 2016 |
Graphene cracks the glass corrosion problem
(Nanowerk News) Researchers at the Center for Multidimensional Carbon Materials (CMCM), within the Institute for Basic Science (IBS) have demonstrated graphene coating protects glass from corrosion. Their research, published in ACS Nano ("Graphene Coatings as Barrier Layers to Prevent the Water-Induced Corrosion of Silicate Glass"), can contribute to solving problems related to glass corrosion in several industries.
|
 |
| An example of a glass showing cracking and opacity due to corrosion.
|
|
Glass has a high degree of both corrosion and chemical resistance. For this reason it is the primary packaging material to preserve medicines and chemicals. However, over time at high humidity and pH, some glass types corrode. Corroded glass loses its transparency and its strength is reduced. As a result, the corrosion of silicate glass, the most common and oldest form of glass, by water is a serious problem especially for the pharmaceutical, environmental and optical industries, and in particular in hot and humid climates.
|
|
Although there are different types of glass, ordinary glazing and containers are made of silicon dioxide (SiO2), sodium oxide (Na2O) along with minor additives. Glass corrosion begins with the adsorption of water on the glass surface. Hydrogen ions from water then diffuse into the glass and exchange with the sodium ions present on the glass surface. The pH of the water near the glass surface increases, allowing the silicate structure to dissolve.
|
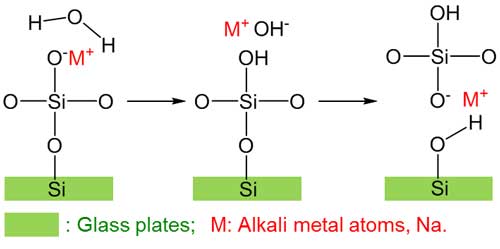 |
| Corrosion mechanism on the glass surface. (© ACS)
|
|
Scientists have been looking at how to coat glass to protect it from damage. An ideal protective coating should be thin, transparent, and provide a good diffusion barrier to chemical attack. Graphene with its chemical inertness, thinness, and high transparency makes it very promising as a coating material. Moreover, owing to its excellent chemical barrier properties it blocks helium atoms from penetrating through it. The use of graphene coating is being explored as a protective layer for other materials requiring resistance to corrosion, oxidation, friction, bacterial infection, electromagnetic radiation, etc.
|
|
IBS scientists grew graphene on copper using a technique previously invented by Prof. Rodney S. Ruoff and collaborators, and transferred either one or two atom-thick layers of graphene onto both sides of rectangular pieces of glass. The effectiveness of the graphene coating was evaluated by water immersion testing and observing the differences between uncoated and coated glass. After 120 days of immersion in water at 60°C, uncoated glass samples had significantly increased in surface roughness and defects, and reduced in fracture strength. In contrast, both the single and double layer graphene-coated glasses had essentially no change in both fracture strength and surface roughness.
|
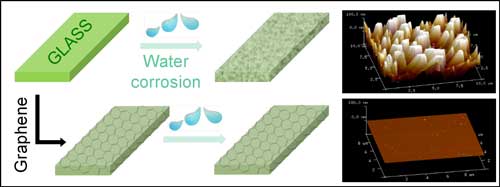 |
| Graphene coatings protect glass from corrosion as shown in the atomic force microscopy images on the right. On uncoated glass (top right), the surface roughness increases after immersion in water for 120 days. In contrast, glass plates coated with graphene films were much more stable and the increase in roughness was negligible after immersion in water for 120 days. (© ACS) (click on image to enlarge)
|
|
“The purpose of the study was to determine whether graphene grown by chemical vapor deposition on copper foils, a now established method, could be transferred onto glass, and protect the glass from corrosion. Our study shows that even one atom-thick layer of graphene does the trick,” explains Prof. Ruoff, director of the CMCM and Professor at the Ulsan National Institute of Science and Technology (UNIST). “In the future, when it is possible to produce larger and yet higher-quality graphene sheets and to optimize the transfer on glass, it seems reasonably likely that graphene coating on glass will be used on an industrial scale.”
|



